Osteosarcoma
 |
Osteogenic Sarcoma (osteosarcoma) is a bone forming cancer. It is the most frequent type of bone tumour and is most common between the ages of 15 to 25. Over 90% of tumours are located in the metaphysis (the growing ends of the bone), the most common sites are the bones around the knee which account for 80% of cases. Osteosarcomas vary greatly in radiological and pathological features and therefore needs careful diagnosis to differentiate this from other bone tumours. Most are high grade intramedullary osteosarcomas, about 5% are low grade lesions, some are secondary osteosarcomas (for example those caused by radiation therapy).
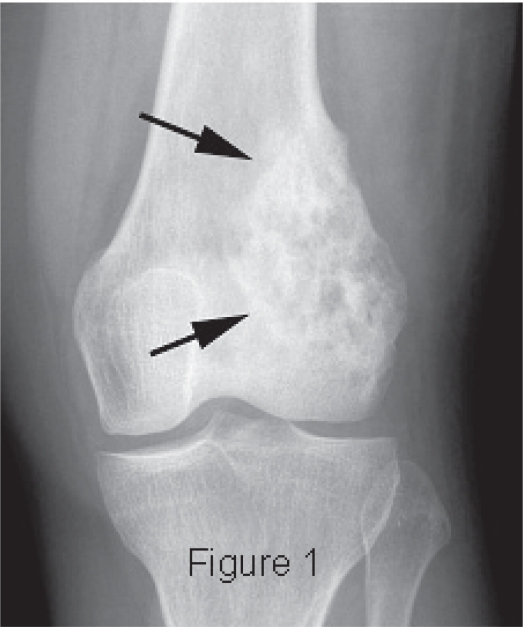
Figure 1. Radiograph showing an osteolytic and osteoblastic intra-medullary tumor characteristic of osteosarcoma. From Layfield J et al. Clin Med Pathol. 2008; 1: 55-59. Available under a Creative Commons CC-BY-3.0 license. |
 Information for Patients and the Public
Information for Patients and the Public Information for Health Professionals / Researchers
Information for Health Professionals / Researchers Latest Research Publications
Latest Research Publications Osteosarcoma FAQs
Osteosarcoma FAQs Bone Cancer Resources
Bone Cancer ResourcesInformation Patients and the Public (13 links)
 Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
National Cancer Institute![]() PDQ summaries are written and frequently updated by editorial boards of experts Further info.
PDQ summaries are written and frequently updated by editorial boards of experts Further info.
NHS Choices![]() NHS Choices information is quality assured by experts and content is reviewed at least every 2 years. Further info.
NHS Choices information is quality assured by experts and content is reviewed at least every 2 years. Further info.
Overview of promary bone cancers in general, though does include some specific information about Osteosarcoma, Ewing's Sarcoma, Chondrosarcoma and Spindle cell sarcoma.
![]() Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
BCRT became a registered the charity in 2006 and raises funds for research into primary bone cancer, and provides information and support for patients and their families. The Website includes information booklets, personal stories and a section for teenagers.
Macmillan Cancer Support![]() Content is developed by a team of information development nurses and content editors, and reviewed by health professionals. Further info.
Content is developed by a team of information development nurses and content editors, and reviewed by health professionals. Further info.
Cancer.Net![]() Content is peer reviewed and Cancer.Net has an Editorial Board of experts and advocates. Content is reviewed annually or as needed. Further info.
Content is peer reviewed and Cancer.Net has an Editorial Board of experts and advocates. Content is reviewed annually or as needed. Further info.
Bone Cancer Research Trust![]() Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Overview of Osteosarcoma and the different types of the disease.
 Osteosarcoma
Osteosarcoma
Mayo Clinic
Dr. Carola Arndt discusses osteosarcoma which is one of the most common malignant tumors of bone in teenagers and young adults. She discusses diagnosis, evaluation, and treatment of osteosarcoma. Also mentions the Children's Oncology group and EURAMOS study. Mentions the multi-disciplinary approach at Mayo Clinic.
 European and American Osteosarcoma Study Group
European and American Osteosarcoma Study Group
EURAMOS
Includes information about the EURAMOS-1 clinical trial. There is also detailed information about osteosarcoma and patients' art work.
 Liddy Shriver Sarcoma Initiative
Liddy Shriver Sarcoma Initiative
Founded in 2003 the initiative aims improve the quality of life for people dealing with sarcomas around the world, raising awareness and research funds. It has an international panel of medical experts.
American Cancer Society
A detailed guides
Childrens' Oncology Group
Includes information, with sections on newly diagnosed, in treatment and after treatment.
A charity for bone and soft-tissue sarcomas in the UK. The organisation raises funds for research and provides information and support.
 Sarcoma UK Online Support Group
Sarcoma UK Online Support Group
Sarcoma UK
This group is for the benefit of sarcoma patients, carers and their families being treated in the UK on the NHS or privately.
Information for Health Professionals / Researchers (9 links)
- PubMed search for publications about Osteosarcoma - Limit search to: [Reviews]
PubMed Central search for free-access publications about Osteosarcoma
MeSH term: Osteosarcoma US National Library of Medicine
US National Library of Medicine PubMed has over 22 million citations for biomedical literature from MEDLINE, life science journals, and online books. Constantly updated.
PubMed has over 22 million citations for biomedical literature from MEDLINE, life science journals, and online books. Constantly updated.
 Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
National Cancer Institute![]() PDQ summaries are written and frequently updated by editorial boards of experts Further info.
PDQ summaries are written and frequently updated by editorial boards of experts Further info.
A website by orthopedic surgeon Dr. Henry DeGroot, with contributions from numerous clinical colleagues. It includes numerous case studies, including radiology and pathology images, and information covering a comprehensive range of bone tumours.
 Case 10: A 12 year old male with an osteolytic lesion of the proximal left tibia
Case 10: A 12 year old male with an osteolytic lesion of the proximal left tibia
Department of Pathology, University of Pittsburgh
Department of Pathology, University of Pittsburgh
 Clinical Case: 51 year old with knee pain (small cell osteosarcoma)
Clinical Case: 51 year old with knee pain (small cell osteosarcoma)
BoneTumour.org
 European and American Osteosarcoma Study Group
European and American Osteosarcoma Study Group
EURAMOS
Includes information about the EURAMOS-1 clinical trial. There is also detailed information about osteosarcoma and patients' art work.
Medscape
A detailed clinical case covering an overview, workup/investigations and treatment and follow-up. The case includes radiology images.
Medscape
A detailed clinical case covering an overview, workup/investigations and treatment. The case includes radiology and pathology images.
 Genetic features of Osteosarcoma
Genetic features of OsteosarcomaLatest Research Publications
This list of publications is regularly updated (Source: PubMed).
A Novel Orthotopic Mouse Model of Lung Metastasis Using Fluorescent Patient-derived Osteosarcoma Cells.
Anticancer Res. 2021; 41(2):635-640 [PubMed] Related Publications
MATERIALS AND METHODS: The OS-PDCs were transfected with GFP-lentivirus. GFP-expressing OS-PDCs (2.0×10
RESULTS: Primary orthotopic tumors were established in two out of three mice. The GFP-expressing OS-PDCs in the PDOC model were visualized. Multiple GFP-expressing lung metastases were detected in one of the two mice with primary tumor.
CONCLUSION: The present study proves the concept that a GFP-expressing PDOC model can mimic clinical lung-metastatic osteosarcoma. This model can serve as a paradigm to screen for effective drugs for osteosarcoma lung metastasis.
Reconstruction using a frozen autograft for a skull and humeral lesion of synchronous multicentric osteosarcoma after undergoing successful neoadjuvant chemotherapy: a case report and review of the literature.
BMC Surg. 2021; 21(1):56 [PubMed] Free Access to Full Article Related Publications
CASE PRESENTATION: We report an 18-year-old girl with SMOS, with lesions located in the left distal femur, right proximal humerus, and left occipital bone. Her major complaint was pain and swelling around the left knee joint. Asymptomatic lesions of the humerus and skull bone were detected on a systemic bone scan. No visceral organ metastasis was observed. A biopsy of the distal femoral lesion revealed osteosarcoma. Based on the histological findings, multiple bone lesions, and absence of visceral lesion, the clinical diagnosis of SMOS was made. After five courses of neoadjuvant chemotherapy with a regimen of doxorubicin and cisplatin, reconstruction using a tumor prosthesis following wide excision of the left distal femur was performed, and total necrosis was histologically observed in the retracted specimen. Following three cycles of adjuvant chemotherapy, tumor excision and reconstruction with a frozen autograft treated with liquid nitrogen was conducted for both lesions of the humerus and skull, rather than tumor prosthesis or synthetics, in order to retain a normal shoulder function, and to obtain a good cosmetic and functional outcome after treatment of the skull lesion. Further adjuvant chemotherapy could not be administered after the completion of the surgical treatment for all lesions because the adverse events due to chemotherapy were observed. At over 5 years after the diagnosis, she remains clinically disease-free.
CONCLUSIONS: An early correct diagnosis, the proper management of chemotherapy, and surgical treatment for all lesions are essential for achieving a good clinical outcome, even in SMOS including a skull lesion. By performing reconstruction using a frozen autograft for a proximal humeral lesion and a skull lesion after confirming the good histological efficacy of neoadjuvant chemotherapy for the primary lesion, the excellent function of the shoulder joint and a good cosmetic outcome at the site of the skull lesion was acquired without complications or recurrence.
Down-regulation of HCP5 inhibits cell proliferation, migration, and invasion through regulating EPHA7 by competitively binding miR-101 in osteosarcoma.
Braz J Med Biol Res. 2021; 54(2):e9161 [PubMed] Free Access to Full Article Related Publications
STAT3 and its targeting inhibitors in osteosarcoma.
Cell Prolif. 2021; 54(2):e12974 [PubMed] Free Access to Full Article Related Publications
A DNA methylation-associated nomogram predicts the overall survival of osteosarcoma.
Medicine (Baltimore). 2020; 99(51):e23772 [PubMed] Free Access to Full Article Related Publications
MXene Surface on Multiple Junction Triangles for Determining Osteosarcoma Cancer Biomarker by Dielectrode Microgap Sensor.
Int J Nanomedicine. 2020; 15:10171-10181 [PubMed] Free Access to Full Article Related Publications
Materials and Methods: This work was focused on diagnosing osteosarcoma (OS), a common bone cancer, on MXene-modified multiple junction triangles by dielectrode sensing. Survivin protein gene is highly correlated with OS, identified on this sensing surface. Capture DNA was immobilized on MXene by using 3-glycidoxypropyltrimethoxysilane as an amine linker and duplexed by the target DNA sequence.
Results: The limitation and sensitivity of detection were found as 1 fM with the acceptable regression co-efficient value (y=1.0037⨰ + 0.525; R
Conclusion: This microgap device with Mxene-modified multiple junction triangles dielectrode surface is beneficial to quantify the survivin gene at its lower level and diagnosing OS complication levels.
Single-cell RNA landscape of intratumoral heterogeneity and immunosuppressive microenvironment in advanced osteosarcoma.
Nat Commun. 2020; 11(1):6322 [PubMed] Free Access to Full Article Related Publications
Comparative analysis of the levels of soluble forms of receptor and ligand of the immunity control point PD-1/PD-L1 in the blood serum of patients with typical bone osteosarcoma and chondrosarcoma.
Klin Lab Diagn. 2020; 65(11):669-675 [PubMed] Related Publications
Mechanism of the Notch signaling pathway in enhancing the efficacy of chemotherapy drugs in osteosarcoma.
Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2020; 45(10):1234-1240 [PubMed] Related Publications
Co-expression of Epstein-Barr virus-encoded RNA1 and viral latent membrane protein 1 in osteosarcoma: A novel insight of predictive markers.
Tumour Biol. 2020; 42(11):1010428320974247 [PubMed] Related Publications
IGFBP-3 stimulates human osteosarcoma cell migration by upregulating VCAM-1 expression.
Life Sci. 2021; 265:118758 [PubMed] Related Publications
MATERIALS AND METHODS: Using a human osteosarcoma tissue array, immunohistochemical staining determined levels of IGFBP-3 expression in osteosarcoma tissue and in normal tissue. The wound healing migration assay, Transwell migration assay, luciferase reporter assay, immunofluorescence staining, Western blot and real-time quantitative PCR were performed to examine whether IGFBP-3 facilitates VCAM-1-dependent migration of osteosarcoma cells.
KEY FINDINGS: In this study, we found significantly higher IGFBP-3 levels in osteosarcoma tissue compared with normal healthy tissue. IGFBP-3 treatment of two human osteosarcoma cell lines promoted cell migration and upregulated levels of VCAM-1 expression via PI3K/Akt and AP-1 signaling.
SIGNIFICANCE: IGFBP-3 appears to be a novel therapeutic target in metastatic osteosarcoma.
Establishment of immune prognostic signature and analysis of prospective molecular mechanisms in childhood osteosarcoma patients.
Medicine (Baltimore). 2020; 99(46):e23251 [PubMed] Free Access to Full Article Related Publications
METHODS: Firstly, we identified immune-related genes in CCLs and CCRs, these genes were used for functional annotation and interaction analysis. Then, the prognostic value of these genes was evaluated using Kaplan-Meier analysis and multivariate COX regression model. And the potential relationship between risk score and immune infiltrating cells was identified. Finally, gene set enrichment analysis was used to determine the underlying molecular mechanism of OS. Immune-related genes in CCLs and CCRs are inextricably linked.
RESULTS: The results of survival analysis of these genes show that CCL5, CCL8, CCR4, and CCR5 are significantly associated with the prognosis of childhood OS. The combined effect survival analysis shows that the co-high expression of these 4 genes has a good prognosis for childhood OS. A prognostic signature model was constructed based on the 4 genes mentioned above, and the result of time-dependent receiver operating characteristic curves showed that this model was a good predictor of childhood OS 3- and 5-year prognosis. In addition, the risk score of the constructed prognostic signature model was closely related to immune infiltration. We also found that CCL5, CCL8, and CCR5 may affect the prognosis of OS through complex regulation among Toll-like receptor signaling pathway, mitogen-activated protein kinase (MAPK) family signaling cascade, and nuclear factor-kappaB pathway, whereas CCR4 affects the prognosis of OS by regulating eukaryotic translation.
CONCLUSION: CCL5, CCL8, CCR4, and CCR5 are potential prognostic markers for the prognosis of childhood OS, and the underlying molecular mechanisms of childhood OS have been identified.
Neuropilin and tolloid-like 2 regulates the progression of osteosarcoma.
Gene. 2021; 768:145292 [PubMed] Related Publications
Different Pattern of Bone Scintigraphy in Mandibular Osteosarcoma Arising From Fibrous Dysplasia in a Patient With McCune-Albright Syndrome.
Clin Nucl Med. 2021; 46(1):e11-e12 [PubMed] Related Publications
Punicalagin suppresses osteosarcoma growth and metastasis by regulating NF-κB signaling.
J Biol Regul Homeost Agents. 2020 Sep-Oct; 34(5):1699-1708 [PubMed] Related Publications
Osteosarcoma of the jaws: An overview of the pathophysiological mechanisms.
Crit Rev Oncol Hematol. 2020; 156:103126 [PubMed] Related Publications
An update on the roles of circular RNAs in osteosarcoma.
Cell Prolif. 2021; 54(1):e12936 [PubMed] Free Access to Full Article Related Publications
Circ-XPO1 upregulates XPO1 expression by sponging multiple miRNAs to facilitate osteosarcoma cell progression.
Exp Mol Pathol. 2020; 117:104553 [PubMed] Related Publications
MicroRNA-761 suppresses tumor progression in osteosarcoma via negatively regulating ALDH1B1.
Life Sci. 2020; 262:118544 [PubMed] Related Publications
MATERIALS AND METHODS: qRT-PCR assay was applied to detect miR-761 expression. CCK-8, colony formation and EdU assays were conducted to explore the functional role of miR-761/ALDH1B1 axis in osteosarcoma. Bioinformatics analysis and luciferase reporter assay was utilized to assess the regulation between miR-761 and ALDH1B1. Mechanism experiments were implemented to investigate the underlying molecular mechanism of miR-761/ALDH1B1 axis.
KEY FINDINGS: ALDH1B1 was negatively regulated by microRNA-761 (miR-761). Functionally, miR-761 suppressed cell growth, migration, and invasion in osteosarcoma via targeting ALDH1B1 in vitro. Xenograft tumor model demonstrated that miR-761 inhibited osteosarcoma tumor development in vivo through regulating ALDH1B1. Consistently, we showed that miR-761 expression was decreased in osteosarcoma patients and low expression of miR-761 was correlated with worse prognosis in osteosarcoma patients. Mechanistically, we revealed that high expression of ALDH1B1 was significantly associated with enhanced TGF-β signaling, epithelial-mesenchymal transition (EMT), and cell adhesion. Furthermore, miR-761 regulated TGF-β and EMT/cell adhesion in osteosarcoma via targeting ALDH1B1.
SIGNIFICANCE: Taken together, our findings suggest that the oncogenic ALDH1B1 is regulated by miR-761 during osteosarcoma development and progression, which might provide a novel prognostic biomarker and therapeutic strategy for osteosarcoma treatment.
Automated Classification of Osteosarcoma and Benign Tumors using RNA-seq and Plain X-ray.
Annu Int Conf IEEE Eng Med Biol Soc. 2020; 2020:1165-1168 [PubMed] Related Publications
Sulforaphene inhibits the progression of osteosarcoma via regulating FSTL1/NF-κB pathway.
Life Sci. 2020; 263:118485 [PubMed] Related Publications
MAIN METHODS: We explored the therapeutic effects of SFE in modulating the progression of osteosarcoma. CCK8 assay, colony formation assay, western blot, wounding healing assay and transwell assay were conducted to detect the proliferation, apoptosis, migration and invasion of osteosarcoma cells (U2OS and Saos2) treated with different concentrations of SFE. In addition, tumor xenograft in nude mice is performed to test the effects of SFE in tumorigenesis in vivo. Moreover, the levels of FSTL1 and NF-κB were determined by western blot, and loss of functions of FATL1 and NF-κB were further conducted to evaluate the underlying mechanisms of SFE on osteosarcoma development.
KEY FINDINGS: The results revealed that SFE inhibited the growth while promoted apoptosis of U2OS and Saos2 cells in a dose-dependent manner. Mechanistically, SFE significantly inhibited the expression of NF-κB and FSTL1. However, the genetic intervention of FSTL1 or pharmacologically inhibiting NF-κB weakened the anti-tumor role of SFE.
SIGNIFICANCE: This study suggested that SFE alleviates the progression of osteosarcoma through modulating the FSTL1/NF-κB pathway.
The effects of common variants in MDM2 and GNRH2 genes on the risk and survival of osteosarcoma in Han populations from Northwest China.
Sci Rep. 2020; 10(1):15939 [PubMed] Free Access to Full Article Related Publications
Heme oxygenase-1 in osteosarcoma.
J Cancer Res Ther. 2020 Jul-Sep; 16(4):874-877 [PubMed] Related Publications
Materials and Methods: Twenty five histopathologically confirmed cases of osteosarcoma localized without metastasis of all the ages attending the Orthopedic Clinics were included in the study group and twenty five patients having musculoskeletal pain (age and sex matched) served as control. Five ml of venous blood was collected aseptically from antecubital vein and serum was be separated by centrifugation and analyzed the same day. Routine biochemistry investigations were performed as per standard enzymatic methods by autoanalyzer. Serum Heme oxygenase-1 was analyzed by enzyme-linked immunosorbent assay.
Results: In osteosarcoma patients, serum HO-1 levels were increased as compared to patients having musculoskeletal pain (P < 0.05). Workers have found that HO-1 induction in prostate cancer cell lines (PC3) cells restored the proliferation of osteoblasts, which was inhibited during co-culture with parental prostate cancer cell line PC3 cells. However, no concrete data are available on blood levels of HO in osteosarcoma. Major role of HO-1 is the protection against oxidative injury, additionally, it regulates cell proliferation, modulates inflammatory response and facilitates angiogenesis.
Conclusion: Findings of the present study suggests that pharmacological agents that regulate HO activity or HO-1 gene silencing may become powerful tools for preventing the onset or progression of various cancers and sensitize them to anticancer therapies.
Extended analysis of intratumoral heterogeneity of primary osteosarcoma tissue using 3D-in-vivo-tumor-model.
Clin Hemorheol Microcirc. 2020; 76(2):133-141 [PubMed] Related Publications
OBJECTIVE: Objective was to establish the grafting and transplantation of different primary osteosarcoma tissue parts onto several consecutive CAMs for tumor profiling and investigation of osteosarcoma heterogeneity.
METHODS: Various parts of primary osteosarcoma tissue were grafted onto CAMs and were transplanted onto another CAM for five to seven consecutive times, enabling further experimental analyzes.
RESULTS: Primary osteosarcoma tissue parts exhibited satisfactory growth patterns and displayed angiogenic development on the CAM. It was possible to graft and transplant different tumor parts several times while the tissue viability was still high and tumor profiling was performed.
CONCLUSIONS: Primary osteosarcoma tissue grew on several different CAMs for an extended time period and neovascularization of serial transplanted tumor parts was observed, improving the versatility of the 3D-in-vivo-tumor-model.
Expression and clinical implications of leucine-rich repeat containing 15 (LRRC15) in osteosarcoma.
J Orthop Res. 2020; 38(11):2362-2372 [PubMed] Related Publications
The impact of miR-9 in osteosarcoma: A study based on meta-analysis, TCGA data, and bioinformatics analysis.
Medicine (Baltimore). 2020; 99(35):e21902 [PubMed] Free Access to Full Article Related Publications
Definitive surgery of primary lesion should be prioritized over preoperative chemotherapy to treat high-grade osteosarcoma in patients aged 41-65 years.
J Orthop Traumatol. 2020; 21(1):13 [PubMed] Free Access to Full Article Related Publications
MATERIALS AND METHODS: We retrospectively analyzed the outcomes of osteosarcoma, especially in regards to preoperative chemotherapy, from January 1980 to July 2014. A total of 29 patients with high-grade osteosarcoma between the age of 40 and 65 years were included. We included patients without distant metastasis and with primary lesions that were deemed resectable. The mean age was 52.8 years (range 41-65 years), and the mean follow-up period was 103.2 months (range 5-314 months).
RESULTS: Adjuvant chemotherapy was administered to 27 of 29 patients (93%), and 8 of 15 cases (53%) were able to undergo preoperative chemotherapy as planned, including CDDP. A major complication of chemotherapy was acute kidney injury due to CDDP (26%). The 5-year OS and 5-year EFS were 64.9% and 57.1%, respectively. After 2006, a policy to prioritize the resection of the primary lesion was implemented, and if the primary lesion was deemed resectable, preoperative chemotherapy was either not administered or administered for only a short duration. The 5-year OS after 2006 improved to 78.8%.
CONCLUSIONS: This study shows that administration of high-dose intensity preoperative chemotherapy was difficult in middle-aged and older patients due to their high rate of acute kidney injury by CDDP. For cases of osteosarcoma in middle-aged and older patients, if the primary lesion is resectable, preoperative chemotherapy should be minimized to prioritize the resection of the primary lesion. It was considered that, with appropriate measures to prevent complications, adjuvant chemotherapy may lead to improved prognosis.
LEVEL OF EVIDENCE: V.
Exploring the association mechanism between metastatic osteosarcoma and non-metastatic osteosarcoma based on dysfunctionality module.
J BUON. 2020 May-Jun; 25(3):1569-1578 [PubMed] Related Publications
METHODS: Differentially expressed genes in metastatic osteosarcoma and non-metastatic osteosarcoma were obtained first. Secondly, co-expression analysis has been processed for differentially expressed genes, and it is necessary to figure the gene drive of each module. Furthermore, both GO function and KEGG pathway enrichment analysis were performed on the module genes. Comprehensively, the module gene set which was predicted according to hypergeometric testing was importantly regulated by both transcription factors (TFs) and non-coding RNAs (ncRNAs).
RESULTS: Conclusively, 16 co-expression modules were obtained. ACAT1 and ATBF1 would actively regulate in dysfunction modules, and thus they are identified as osteosarcoma-driven genes. Enrichment results showed that the module genes were significantly involved in transcription factor activity, specific DNA binding of the RNA polymerase II proximal promoter sequence, DNA-binding transcriptional activator activity, ubiquitin-like protein transferase activity, and another biological process. Moreover, module genes significantly regulates FcγR-mediated phagocytosis, MAPK signaling pathway, phagocytosis, PI3K-Akt signaling pathway and others. Finally, we identified pivot ncRNAs (including CRNDE, miR-106a-5p, miR-181a-5p, etc) and pivot TFs (including NFKB1, STAT6, PPARG, RELA, etc) that significantly regulate dysfunction modules.
CONCLUSION: Overall, this work deciphered a co-expression network of common core pathogenic genes including metastatic osteosarcoma and non-metastatic osteosarcoma. It helps to identify core dysfunction modules and potential regulatory factors of the disease and improves understanding the underlying molecular association mechanisms between the two diseases.
Effects of miR-432 and miR-548c-3p on the proliferation and invasion of osteosarcoma cells.
J BUON. 2020 May-Jun; 25(3):1562-1568 [PubMed] Related Publications
METHODS: A total of 67 cases of patients with osteosarcoma who came to the third Affiliated Hospital of Southern Medical University from April 2015 to May 2018 formed the experimental group, and 63 healthy individuals who came to this hospital for physical examination during the same period formed the control group. The expressions of miR-432 and miR-548c-3p in sera of each group were detected by RT-PCR to observe the changes of the expression levels of these miRs in the sera of the experimental and the control group, and the relationship between the expression levels of these miRs in the sera of patients with osteosarcoma and the grade of tumor differentiation and different pathological classification. GM-63 cells were selected as the target for in vitro experiments which were cultured and transfected. Before transfection, cells were divided into blank group (without transfection), negative control group (transfected with miR NC) and experimental group (transfected with miR-432 mimics/miR-548c-3p mimics). Cell proliferation was detected by CCK-8 method, cell apoptosis was detected by flow cytometry, and cell invasion by transwell invasion experiment.
RESULTS: miR-423 and miR-548c-3p showed low expression in osteosarcoma, and the overexpression of miR-432 and miR-548c-3p in osteosarcoma cells could inhibit the proliferation of tumor cells and promote their apoptosis. miR-432 and miR-548c-3p might be tumor suppressors of osteosarcoma, and their expression levels could be used as important reference indexes to evaluate the benign and malignant levels of osteosarcoma.
CONCLUSION: These results suggested that the abnormal expressions of miR-432 and miR-548c-3p may be key factors of the occurrence and development of osteosarcoma.
MEX3C promotes osteosarcoma malignant progression through negatively regulating FGF14.
J BUON. 2020 May-Jun; 25(3):1554-1561 [PubMed] Related Publications
METHODS: MEX3C levels in tumor tissues and adjacent ones of 52 OS patients were studied by quantitative real-time polymerase chain reaction (qRT-PCR), and the relationship between MEX3C expression and clinicopathological characteristics of OS patients was analyzed. At the same time, qRT-PCR further verified the MEX3C level in OS cell lines, and HOS and MG63 OS cell lines were selected to construct MEX3C overexpression and knockdown cell model, respectively. The impact of MEX3C on OS cell functions were determined by cell wound healing and transwell assay. In addition, the interaction between MEX3C and FGF14 was further determined by luciferase assay, western blot and recovery experiments.
RESULTS: MEX3C had increased expression both in OS tissue samples and in OS cell lines. High expression of MEX3C was predictive of high incidence of nodal involvement or distant metastasis. Silencing MEX3C remarkably attenuated the migration ability of OS cells, while, conversely, overexpression enhanced that. Bioinformatics analysis and luciferase assay confirmed that MEX3C bind to FGF14 directly, and the expression of FGF14 was significantly reduced in OS tumor tissue specimens, and was negatively correlated with MEX3C. Overexpression of FGF14 was able to reverse the promoting effect of MEX3C on the crawling ability and invasiveness of OS cells.
CONCLUSIONS: MEX3C was remarkably increased in OS tissues and was remarkably correlated with the incidence of metastasis of OS patients. In addition, MEX3C accelerated the malignant progression of OS through negatively modulating FGF14.
 Bone Cancers
Bone Cancers