Found this page useful?
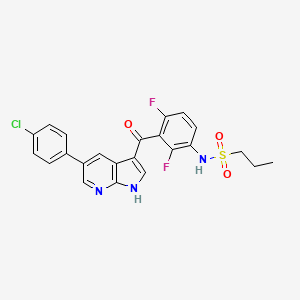
Vemurafenib (Zelboraf)
 Web Resources: Vemurafenib (Zelboraf)
Web Resources: Vemurafenib (Zelboraf) Latest Research Publications: Vemurafenib
Latest Research Publications: VemurafenibWeb Resources: Vemurafenib (Zelboraf) (6 links)
American Cancer Society
MedlinePlus.gov
NHS Evidence
 Vemurafenib - Substance Summary
Vemurafenib - Substance Summary
PubChem
Macmillan Cancer Support
Cancer Research UK
Latest Research Publications: Vemurafenib
Schaefer T, Satzger I, Gutzmer R
Clinics, prognosis and new therapeutic options in patients with mucosal melanoma: A retrospective analysis of 75 patients.
Medicine (Baltimore). 2017; 96(1):e5753 [PubMed] Free Access to Full Article Related Publications
Clinics, prognosis and new therapeutic options in patients with mucosal melanoma: A retrospective analysis of 75 patients.
Medicine (Baltimore). 2017; 96(1):e5753 [PubMed] Free Access to Full Article Related Publications
Mucosal melanomas represent a rare entity with different risk factors and molecular features compared to cutaneous melanomas. They arise most commonly from mucosal surfaces in the head/neck region, the female genital tract (FGT) and the anorectal region. The aim of this study was to evaluate clinics, prognosis, and treatment options of patients with mucosal melanoma, in particular with regard to different primary sites.We retrospectively analyzed 75 patients with mucosal melanomas diagnosed in the years 1993 to 2015 in our department. The primary melanomas were located in the head/neck region (n = 32), the FGT (n = 24), and the anorectal region (n = 19).The median age of the patients was 66 years. At initial diagnosis the primary melanoma was not completely resectable in 11 (15%) patients, 18 (24%) patients had regional lymph node metastases, and 7 (9%) patients distant metastases. During follow-up, 22 (29%) patients suffered from a local recurrence, in particular patients with primary melanoma in the head/neck region without postoperative radiotherapy. By multivariate analysis location of the primary melanoma in the head/neck area or anorectal region and presence of metastases at time of diagnosis represented poor prognostic factors for recurrence-free survival. In 62 tested individuals 7 KIT mutations were found, 2 BRAF mutations in 57 tested patients. Four patients received targeted therapies, 14 checkpoint inhibitors, 4 (1/1 on vemurafenib, 1/7 on ipilimumab, and 2/7 on PD-1 inhibitors) patients showed responses of more than 100 days duration.Mucosal melanomas are often locally advanced or metastatic at initial diagnosis, thus they require extensive staging procedures. The high rate of local recurrences in the head/neck region can be significantly reduced by postoperative radiotherapy. For the potential use of medical treatment a mutation analysis for KIT and BRAF genes should be performed. The use of new immunologic and targeted therapies has to be further evaluated.
Simeone E, Grimaldi AM, Festino L, et al.
Combination Treatment of Patients with BRAF-Mutant Melanoma: A New Standard of Care.
BioDrugs. 2017; 31(1):51-61 [PubMed] Related Publications
Combination Treatment of Patients with BRAF-Mutant Melanoma: A New Standard of Care.
BioDrugs. 2017; 31(1):51-61 [PubMed] Related Publications
Raf-mitogen-activated protein kinase (Raf-MAPK) pathway inhibition with the BRAF inhibitors vemurafenib and dabrafenib, alone or in combination with a MEK inhibitor, has become a standard therapeutic approach in patients with BRAF-mutated metastatic melanoma. Both vemurafenib and dabrafenib have shown good safety and efficacy as monotherapy compared with chemotherapy. However, the duration of response is limited in the majority of patients treated with BRAF inhibitor monotherapy because of the development of acquired resistance. The addition of a MEK inhibitor can improve blockade of the MAPK pathway and may help to overcome resistance and thereby prolong efficacy, as well as reduce cutaneous toxicity. Combinations of BRAF inhibitors and MEK inhibitors (dabrafenib plus trametinib and vemurafenib plus cobimetinib) have been approved for the treatment of BRAF-mutant metastatic melanoma and may become a new standard of care. However, acquired resistance is still a significant concern with BRAF and MEK inhibitor combination therapy, and other strategies are being investigated, including the use of sequential and intermittent schedules. The combination of BRAF or MEK inhibitors with immunotherapy has been shown to hold considerable promise, with several combinations being evaluated in clinical trials. Preliminary results from clinical trials involving triple combination therapy with BRAF-MEK inhibitors and anti-PD-L1 antibodies appear promising and may indicate a new strategy to treat patients with BRAF-mutated metastatic melanoma. Biomarkers are needed to help identify patients with BRAFV600 mutations most likely to benefit from first-line BRAF/MEK inhibitor therapy rather than immunotherapy and vice versa.
Kroeze SG, Fritz C, Hoyer M, et al.
Toxicity of concurrent stereotactic radiotherapy and targeted therapy or immunotherapy: A systematic review.
Cancer Treat Rev. 2017; 53:25-37 [PubMed] Related Publications
Toxicity of concurrent stereotactic radiotherapy and targeted therapy or immunotherapy: A systematic review.
Cancer Treat Rev. 2017; 53:25-37 [PubMed] Related Publications
BACKGROUND AND PURPOSE: Both stereotactic radiotherapy (SRT) and immune- or targeted therapy play an increasingly important role in personalized treatment of metastatic disease. Concurrent application of both therapies is rapidly expanding in daily clinical practice. In this systematic review we summarize severe toxicity observed after concurrent treatment.
MATERIAL AND METHODS: PubMed and EMBASE databases were searched for English literature published up to April 2016 using keywords "radiosurgery", "local ablative therapy", "gamma knife" and "stereotactic", combined with "bevacizumab", "cetuximab", "crizotinib", "erlotinib", "gefitinib", "ipilimumab", "lapatinib", "sorafenib", "sunitinib", "trastuzumab", "vemurafenib", "PLX4032", "panitumumab", "nivolumab", "pembrolizumab", "alectinib", "ceritinib", "dabrafenib", "trametinib", "BRAF", "TKI", "MEK", "PD1", "EGFR", "CTLA-4" or "ALK". Studies performing SRT during or within 30days of targeted/immunotherapy, reporting severe (⩾Grade 3) toxicity were included.
RESULTS: Concurrent treatment is mostly well tolerated in cranial SRT, but high rates of severe toxicity were observed for the combination with BRAF-inhibitors. The relatively scarce literature on extra-cranial SRT shows a potential risk of increased toxicity when SRT is combined with EGFR-targeting tyrosine kinase inhibitors and bevacizumab, which was not observed for cranial SRT.
CONCLUSIONS: This review gives a best-possible overview of current knowledge and its limitations and underlines the need for a timely generation of stronger evidence in this rapidly expanding field.
MATERIAL AND METHODS: PubMed and EMBASE databases were searched for English literature published up to April 2016 using keywords "radiosurgery", "local ablative therapy", "gamma knife" and "stereotactic", combined with "bevacizumab", "cetuximab", "crizotinib", "erlotinib", "gefitinib", "ipilimumab", "lapatinib", "sorafenib", "sunitinib", "trastuzumab", "vemurafenib", "PLX4032", "panitumumab", "nivolumab", "pembrolizumab", "alectinib", "ceritinib", "dabrafenib", "trametinib", "BRAF", "TKI", "MEK", "PD1", "EGFR", "CTLA-4" or "ALK". Studies performing SRT during or within 30days of targeted/immunotherapy, reporting severe (⩾Grade 3) toxicity were included.
RESULTS: Concurrent treatment is mostly well tolerated in cranial SRT, but high rates of severe toxicity were observed for the combination with BRAF-inhibitors. The relatively scarce literature on extra-cranial SRT shows a potential risk of increased toxicity when SRT is combined with EGFR-targeting tyrosine kinase inhibitors and bevacizumab, which was not observed for cranial SRT.
CONCLUSIONS: This review gives a best-possible overview of current knowledge and its limitations and underlines the need for a timely generation of stronger evidence in this rapidly expanding field.
Zhao Y, Wang W, Min I, et al.
BRAF V600E-dependent role of autophagy in uveal melanoma.
J Cancer Res Clin Oncol. 2017; 143(3):447-455 [PubMed] Related Publications
BRAF V600E-dependent role of autophagy in uveal melanoma.
J Cancer Res Clin Oncol. 2017; 143(3):447-455 [PubMed] Related Publications
BACKGROUND: Autophagy can function in a dual role in cancer development and progression: It can be cytoprotective or contribute to cell death. Therefore, determining the contextual role of autophagy between these two opposing effects is important. So far, little is known about the role of autophagy in uveal melanoma. In the present study, we looked to investigate the autophagic process, as well as its effect on cell survival in uveal melanoma cell lines under stressed conditions (starvation). The possible role of autophagy during BRAF inhibition in uveal melanoma was also sought.
METHODS: Two human uveal melanoma cell lines, OCM1A, which harbors the BRAF mutation V600E and Mel 290, which is BRAF wild type, were studied. Autophagy levels were determined by Western blot assay with/without the addition of autophagic flux inhibitor (bafilomycin A1). Cell proliferation was assessed by an MTT assay.
RESULTS: Starvation triggered autophagy in BRAF V600E-mutant OCM1A cells but not in BRAF wild-type Mel 290 cells. Enhanced autophagy helped the OCM1A cells survive under stressed conditions. The BRAF inhibitor vemurafenib upregulated autophagy through suppression of the PI3K/Akt/mTOR/p70S6 K pathway in BRAF V600E-mutant uveal melanoma cells. Autophagy inhibition impaired the treatment efficacy of vemurafenib in BRAF V600E-mutant uveal melanoma cells.
CONCLUSIONS: Our data demonstrate that starvation-trigged autophagy, which is BRAF V600E dependent, promotes cancer cell survival in uveal melanoma. Vemurafenib induces autophagic cell death rather than adaptive cell survival in BRAF V600E-mutant melanoma.
METHODS: Two human uveal melanoma cell lines, OCM1A, which harbors the BRAF mutation V600E and Mel 290, which is BRAF wild type, were studied. Autophagy levels were determined by Western blot assay with/without the addition of autophagic flux inhibitor (bafilomycin A1). Cell proliferation was assessed by an MTT assay.
RESULTS: Starvation triggered autophagy in BRAF V600E-mutant OCM1A cells but not in BRAF wild-type Mel 290 cells. Enhanced autophagy helped the OCM1A cells survive under stressed conditions. The BRAF inhibitor vemurafenib upregulated autophagy through suppression of the PI3K/Akt/mTOR/p70S6 K pathway in BRAF V600E-mutant uveal melanoma cells. Autophagy inhibition impaired the treatment efficacy of vemurafenib in BRAF V600E-mutant uveal melanoma cells.
CONCLUSIONS: Our data demonstrate that starvation-trigged autophagy, which is BRAF V600E dependent, promotes cancer cell survival in uveal melanoma. Vemurafenib induces autophagic cell death rather than adaptive cell survival in BRAF V600E-mutant melanoma.
Luo C, Lim JH, Lee Y, et al.
A PGC1α-mediated transcriptional axis suppresses melanoma metastasis.
Nature. 2016; 537(7620):422-426 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
A PGC1α-mediated transcriptional axis suppresses melanoma metastasis.
Nature. 2016; 537(7620):422-426 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Melanoma is the deadliest form of commonly encountered skin cancer because of its rapid progression towards metastasis. Although metabolic reprogramming is tightly associated with tumour progression, the effect of metabolic regulatory circuits on metastatic processes is poorly understood. PGC1α is a transcriptional coactivator that promotes mitochondrial biogenesis, protects against oxidative stress and reprograms melanoma metabolism to influence drug sensitivity and survival. Here, we provide data indicating that PGC1α suppresses melanoma metastasis, acting through a pathway distinct from that of its bioenergetic functions. Elevated PGC1α expression inversely correlates with vertical growth in human melanoma specimens. PGC1α silencing makes poorly metastatic melanoma cells highly invasive and, conversely, PGC1α reconstitution suppresses metastasis. Within populations of melanoma cells, there is a marked heterogeneity in PGC1α levels, which predicts their inherent high or low metastatic capacity. Mechanistically, PGC1α directly increases transcription of ID2, which in turn binds to and inactivates the transcription factor TCF4. Inactive TCF4 causes downregulation of metastasis-related genes, including integrins that are known to influence invasion and metastasis. Inhibition of BRAF(V600E) using vemurafenib, independently of its cytostatic effects, suppresses metastasis by acting on the PGC1α-ID2-TCF4-integrin axis. Together, our findings reveal that PGC1α maintains mitochondrial energetic metabolism and suppresses metastasis through direct regulation of parallel acting transcriptional programs. Consequently, components of these circuits define new therapeutic opportunities that may help to curb melanoma metastasis.
Powell MR, Sheehan DJ, Kleven DT
Altered Morphology and Immunohistochemical Characteristics in Metastatic Malignant Melanoma After Therapy With Vemurafenib.
Am J Dermatopathol. 2016; 38(9):e137-9 [PubMed] Related Publications
Altered Morphology and Immunohistochemical Characteristics in Metastatic Malignant Melanoma After Therapy With Vemurafenib.
Am J Dermatopathol. 2016; 38(9):e137-9 [PubMed] Related Publications
Metastatic melanoma is traditionally diagnosed using classic morphologic features in addition to immunohistochemical studies. The authors report a case of metastatic malignant melanoma where both morphology and immunohistochemistry were altered after treatment. This 51-year-old patient presented with metastatic melanoma to the brain and axilla. Initially, both metastases showed classic morphology and diffuse staining with the pan-melanoma antibody cocktail. This cocktail is a combination of 3 antibodies commonly used to diagnose melanocytic neoplasms: Melan-A (MART-1), tyrosinase, and HMB-45. In combination, the cocktail is highly sensitive for detecting melanocytic neoplasms and is commonly used to diagnose metastatic melanoma. Her tumor was positive for the BRAF 1799T>A (V600E) mutation, and she was treated with BRAF inhibitor therapy (vemurafenib). However, the axillary tumor recurred after treatment with vemurafenib. The recurrent tumor showed a markedly different morphology and complete loss of staining with the pan-melanoma antibody cocktail. This loss of staining accompanied by the change in morphology was an observation not previously documented after therapy with vemurafenib. This case demonstrates a potential pitfall in the diagnosis of metastatic or recurrent malignant melanoma.
Erfan G, Puig S, Carrera C, et al.
Development of Cutaneous Toxicities During Selective Anti-BRAF Therapies: Preventive Role of Combination with MEK Inhibitors.
Acta Derm Venereol. 2017; 97(2):258-260 [PubMed] Related Publications
Development of Cutaneous Toxicities During Selective Anti-BRAF Therapies: Preventive Role of Combination with MEK Inhibitors.
Acta Derm Venereol. 2017; 97(2):258-260 [PubMed] Related Publications
is missing (Short communication).
Behling F, Barrantes-Freer A, Skardelly M, et al.
Frequency of BRAF V600E mutations in 969 central nervous system neoplasms.
Diagn Pathol. 2016; 11(1):55 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Frequency of BRAF V600E mutations in 969 central nervous system neoplasms.
Diagn Pathol. 2016; 11(1):55 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
BACKGROUND: Treatment options for oncological diseases have been enhanced by the advent of targeted therapies. The point mutation of the BRAF gene at codon 600 (BRAF V600E) is found in several tumor entities and can be approached with selective inhibitory antibodies. The BRAF inhibitor vemurafenib has demonstrated clinical efficacy in patients with BRAF V600E-mutant melanoma brain metastases and in other cancer diseases. Therefore the BRAF V600E mutation is a highly interesting oncological target in brain tumors.
METHODS: This study assesses the BRAF V600E mutation status in 969 intracranial neoplasms using a tissue microarray method and immunohistochemical staining with the mutation-specific VE-1 antibody, followed by sequencing of positively stained cases.
RESULTS: Out of 784 primary brain tumors seven cases with a BRAF V600E mutation were detected (7/784, 1 %). Six of these cases were neuroepithelial tumors (6/667, 1 %) encompassing 2 astrocytomas WHO grade II (2/42, 5 %), 1 gliosarcoma WHO grade IV (1/75, 1 %) and 3 glioblastomas WHO grade IV (3/312, 1 %). Interestingly, all three mutant glioblastomas showed epithelioid histopathological features. Patients with V600E mutated astrocytic tumors were significantly younger (mean age 15.3 years) than wildtype cases (58.2 years). Among three rhabdoid meningiomas, one case was mutated (1/3) while all other grade I-III meningiomas (1/116, 1 %) and all fifty vestibular schwannomas analyzed were of wildtype status. The vast majority of the BRAF V600E mutations were found in cerebral metastases of malignant melanomas and carcinomas (29/135, 22 %), with false-positive staining found in four breast cancer cases and two non-small-cell lung carcinoma (NSCLC) samples.
CONCLUSIONS: Our data suggest routine screening for BRAF V600E mutations for glioblastomas WHO grade IV below the age of 30, especially in glioblastomas with epithelioid features and in all rhabdoid meningiomas WHO grade III. For colorectal carcinoma, thyroid cancer, malignant melanoma and gliomas BRAF V600E immunostaining is sufficient for screening purposes. We also recommend routine immunohistochemical staining followed by sequencing validation in rare CNS metastases or metastases of unknown primary. Immunohistochemical analysis using mutation-specific antibodies on tissue microarrays is a feasible, time- and cost-efficient approach to high-throughput screening for specific mutations in large tumor series but sequencing validation is necessary in unexpected cases.
METHODS: This study assesses the BRAF V600E mutation status in 969 intracranial neoplasms using a tissue microarray method and immunohistochemical staining with the mutation-specific VE-1 antibody, followed by sequencing of positively stained cases.
RESULTS: Out of 784 primary brain tumors seven cases with a BRAF V600E mutation were detected (7/784, 1 %). Six of these cases were neuroepithelial tumors (6/667, 1 %) encompassing 2 astrocytomas WHO grade II (2/42, 5 %), 1 gliosarcoma WHO grade IV (1/75, 1 %) and 3 glioblastomas WHO grade IV (3/312, 1 %). Interestingly, all three mutant glioblastomas showed epithelioid histopathological features. Patients with V600E mutated astrocytic tumors were significantly younger (mean age 15.3 years) than wildtype cases (58.2 years). Among three rhabdoid meningiomas, one case was mutated (1/3) while all other grade I-III meningiomas (1/116, 1 %) and all fifty vestibular schwannomas analyzed were of wildtype status. The vast majority of the BRAF V600E mutations were found in cerebral metastases of malignant melanomas and carcinomas (29/135, 22 %), with false-positive staining found in four breast cancer cases and two non-small-cell lung carcinoma (NSCLC) samples.
CONCLUSIONS: Our data suggest routine screening for BRAF V600E mutations for glioblastomas WHO grade IV below the age of 30, especially in glioblastomas with epithelioid features and in all rhabdoid meningiomas WHO grade III. For colorectal carcinoma, thyroid cancer, malignant melanoma and gliomas BRAF V600E immunostaining is sufficient for screening purposes. We also recommend routine immunohistochemical staining followed by sequencing validation in rare CNS metastases or metastases of unknown primary. Immunohistochemical analysis using mutation-specific antibodies on tissue microarrays is a feasible, time- and cost-efficient approach to high-throughput screening for specific mutations in large tumor series but sequencing validation is necessary in unexpected cases.
Benamar M, Guessous F, Du K, et al.
Inactivation of the CRL4-CDT2-SET8/p21 ubiquitylation and degradation axis underlies the therapeutic efficacy of pevonedistat in melanoma.
EBioMedicine. 2016; 10:85-100 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Inactivation of the CRL4-CDT2-SET8/p21 ubiquitylation and degradation axis underlies the therapeutic efficacy of pevonedistat in melanoma.
EBioMedicine. 2016; 10:85-100 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
UNLABELLED: The cullin-based CRL4-CDT2 ubiquitin ligase is emerging as a master regulator of cell proliferation. CRL4-CDT2 prevents re-initiation of DNA replication during the same cell cycle "rereplication" through targeted degradation of CDT1, SET8 and p21 during S-phase of the cell cycle. We show that CDT2 is overexpressed in cutaneous melanoma and predicts poor overall and disease-free survival. CDT2 ablation inhibited a panel of melanoma cell lines through the induction of SET8- and p21-dependent DNA rereplication and senescence. Pevonedistat (MLN4924), a specific inhibitor of the NEDD8 activating enzyme (NAE), inhibits the activity of cullin E3 ligases, thereby stabilizing a vast number of cullin substrates and resulting in cancer cell inhibition in vitro and tumor suppression in nude mice. We demonstrate that pevonedistat is effective at inhibiting the proliferation of melanoma cell lines in vitro through the induction of rereplication-dependent permanent growth arrest as well as through a transient, non-rereplication-dependent mechanism. CRISPR/Cas9-mediated heterozygous deletion of CDKN1A (encoding p21) or SET8 in melanoma cells demonstrated that the rereplication-mediated cytotoxicity of pevonedistat is mediated through preventing the degradation of p21 and SET8 and is essential for melanoma suppression in nude mice. By contrast, pevonedistat-induced transient growth suppression was independent of p21 or SET8, and insufficient to inhibit tumor growth in vivo. Pevonedistat additionally synergized with the BRAF kinase inhibitor PLX4720 to inhibit BRAF melanoma, and suppressed PLX4720-resistant melanoma cells. These findings demonstrate that the CRL4-CDT2-SET8/p21 degradation axis is the primary target of inhibition by pevonedistat in melanoma and suggest that a broad patient population may benefit from pevonedistat therapy.
RESEARCH IN CONTEXT: The identification of new molecular targets and effective inhibitors is of utmost significance for the clinical management of melanoma. This study identifies CDT2, a substrate receptor for the CRL4 ubiquitin ligase, as a prognostic marker and therapeutic target in melanoma. CDT2 is required for melanoma cell proliferation and inhibition of CRL4(CDT2) by pevonedistat suppresses melanoma in vitro and in vivo through the induction of DNA rereplication and senescence through the stabilization of the CRL4(CDT2) substrates p21 and SET8. Pevonedistat also synergizes with vemurafenib in vivo and suppresses vemurafenib-resistant melanoma cells. These findings show a significant promise for targeting CRL4(CDT2) therapeutically.
RESEARCH IN CONTEXT: The identification of new molecular targets and effective inhibitors is of utmost significance for the clinical management of melanoma. This study identifies CDT2, a substrate receptor for the CRL4 ubiquitin ligase, as a prognostic marker and therapeutic target in melanoma. CDT2 is required for melanoma cell proliferation and inhibition of CRL4(CDT2) by pevonedistat suppresses melanoma in vitro and in vivo through the induction of DNA rereplication and senescence through the stabilization of the CRL4(CDT2) substrates p21 and SET8. Pevonedistat also synergizes with vemurafenib in vivo and suppresses vemurafenib-resistant melanoma cells. These findings show a significant promise for targeting CRL4(CDT2) therapeutically.
Casadevall D, Vidal J, Gallardo F, et al.
Dabrafenib in an elderly patient with metastatic melanoma and BRAF V600R mutation: a case report.
J Med Case Rep. 2016; 10(1):158 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Dabrafenib in an elderly patient with metastatic melanoma and BRAF V600R mutation: a case report.
J Med Case Rep. 2016; 10(1):158 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
BACKGROUND: Approximately 50 % of malignant melanomas harbor activating point mutations in the BRAF gene. Typically, these mutations result in the substitution of the amino acid valine at codon 600 of the gene, and 90-95 % of mutations are either BRAF (V600E) or BRAF (V600K). Specific BRAF inhibitors such as dabrafenib and vemurafenib are the mainstays of treatment in patients with metastatic BRAF-mutant malignant melanomas. The third most common BRAF mutation is V600R, which also leads to increased BRAF signaling. Although evidence exists about the activity of dabrafenib and vemurafenib in patients with the BRAF (V600R) mutation, these patients have been systematically excluded from recent trials with targeted therapies.
CASE PRESENTATION: Here, we report the positive results in terms of survival and quality of life obtained with dabrafenib in an 80-year-old Caucasian male patient with a Charlson Comorbidity Index of 8 diagnosed with metastatic malignant melanoma harboring the BRAF (V600R) mutation. Our patient was treated with dabrafenib for 7 months with minimal toxicity. We also report exploratory analyses of circulating tumor DNA during targeted treatment. Interestingly, the mutation was not detected after starting treatment and became detectable before radiological disease progression.
CONCLUSIONS: Our report suggests that (1) a relevant benefit can be obtained with a BRAF inhibitor in real-world patients with a malignant melanoma harboring a BRAF (V600R) mutation, and that (2) circulating tumor DNA detection might be of help in assessing tumor burden in everyday clinical practice. The results reported here should encourage the inclusion of patients with BRAF (V600R)-mutated malignant melanomas in future prospective clinical trials with BRAF inhibitors.
CASE PRESENTATION: Here, we report the positive results in terms of survival and quality of life obtained with dabrafenib in an 80-year-old Caucasian male patient with a Charlson Comorbidity Index of 8 diagnosed with metastatic malignant melanoma harboring the BRAF (V600R) mutation. Our patient was treated with dabrafenib for 7 months with minimal toxicity. We also report exploratory analyses of circulating tumor DNA during targeted treatment. Interestingly, the mutation was not detected after starting treatment and became detectable before radiological disease progression.
CONCLUSIONS: Our report suggests that (1) a relevant benefit can be obtained with a BRAF inhibitor in real-world patients with a malignant melanoma harboring a BRAF (V600R) mutation, and that (2) circulating tumor DNA detection might be of help in assessing tumor burden in everyday clinical practice. The results reported here should encourage the inclusion of patients with BRAF (V600R)-mutated malignant melanomas in future prospective clinical trials with BRAF inhibitors.
van Akkooi AC, Atkins MB, Agarwala SS, Lorigan P
Surgical Management and Adjuvant Therapy for High-Risk and Metastatic Melanoma.
Am Soc Clin Oncol Educ Book. 2016; 35:e505-14 [PubMed] Related Publications
Surgical Management and Adjuvant Therapy for High-Risk and Metastatic Melanoma.
Am Soc Clin Oncol Educ Book. 2016; 35:e505-14 [PubMed] Related Publications
Wide local excision is considered routine therapy after initial diagnosis of primary melanoma to reduce local recurrences, but it does not impact survival. Sentinel node staging is recommended for melanomas of intermediate thickness, but it has also not demonstrated any indisputable therapeutic effect on survival. The prognostic value of sentinel node staging has been long established and is therefore considered routine, especially in light of the eligibility criteria for adjuvant therapy (trials). Whether completion lymph node dissection after a positive sentinel node biopsy improves survival is the question of current trials. The MSLT-2 study is best powered to show a potential benefit, but it has not yet reported any data. Another study, the German DECOG study, presented at the 2015 American Society of Clinical Oncology (ASCO) Annual Meeting did not show any benefit but is criticized for the underpowered design and insufficient follow-up. There is no consensus on the use of adjuvant interferon in melanoma. This topic has been the focus of many studies with different regimens (low-, intermediate-, or high-dose and/or short- or long-term treatment). Adjuvant interferon has been shown to improve relapse-free survival but failed to improve overall survival. More recently, adjuvant ipilimumab has also demonstrated an improved relapse-free survival. Overall survival data have not yet been reported due to insufficient follow-up. Currently, studies are ongoing to analyze the use of adjuvant anti-PD-1 and molecular targeted therapies (vemurafenib, dabrafenib, and trametinib). In the absence of unambiguously positive approved agents, clinical trial participation remains a priority. This could change in the near future.
Lee D, Porter J, Hertel N, et al.
Modelling Comparative Efficacy of Drugs with Different Survival Profiles: Ipilimumab, Vemurafenib and Dacarbazine in Advanced Melanoma.
BioDrugs. 2016; 30(4):307-19 [PubMed] Related Publications
Modelling Comparative Efficacy of Drugs with Different Survival Profiles: Ipilimumab, Vemurafenib and Dacarbazine in Advanced Melanoma.
BioDrugs. 2016; 30(4):307-19 [PubMed] Related Publications
BACKGROUND: In the absence of head-to-head data, a common method for modelling comparative survival for cost-effectiveness analysis is estimating hazard ratios from trial publications. This assumes that the hazards of mortality are proportional between treatments and that outcomes are not polluted by subsequent therapy use. Newer techniques that compare treatments where the proportional hazards assumption is violated and adjust for use of subsequent therapies often require patient-level data, which are rarely available for all treatments.
OBJECTIVE: The objective of this study was to provide a comparison of overall survival data for ipilimumab, vemurafenib and dacarbazine using data from three trials lacking a common comparator arm and confounded by the use of subsequent treatment.
METHODS: We compared three estimated overall survival curves for vemurafenib and the difference compared to ipilimumab and dacarbazine. We performed a naïve comparison and adjusted it for heterogeneity between the ipilimumab and vemurafenib trials, including differences in prognostic characteristics and subsequent therapy using a published hazard function for the impact of prognostic characteristics in melanoma and trial data on the impact of second-line use of ipilimumab.
RESULTS: The mean incremental life-years gained for patients receiving ipilimumab compared with vemurafenib were 0.34 (95 % confidence interval [CI] -0.24 to 0.84) using the naïve comparison and 0.51 (95 % CI -0.08 to 0.99) using the covariate-adjusted survival curve.
CONCLUSIONS: The analyses estimated the comparative efficacy of ipilimumab and vemurafenib in the absence of head-to-head patient-level data for all trials and proportional hazards in overall survival.
OBJECTIVE: The objective of this study was to provide a comparison of overall survival data for ipilimumab, vemurafenib and dacarbazine using data from three trials lacking a common comparator arm and confounded by the use of subsequent treatment.
METHODS: We compared three estimated overall survival curves for vemurafenib and the difference compared to ipilimumab and dacarbazine. We performed a naïve comparison and adjusted it for heterogeneity between the ipilimumab and vemurafenib trials, including differences in prognostic characteristics and subsequent therapy using a published hazard function for the impact of prognostic characteristics in melanoma and trial data on the impact of second-line use of ipilimumab.
RESULTS: The mean incremental life-years gained for patients receiving ipilimumab compared with vemurafenib were 0.34 (95 % confidence interval [CI] -0.24 to 0.84) using the naïve comparison and 0.51 (95 % CI -0.08 to 0.99) using the covariate-adjusted survival curve.
CONCLUSIONS: The analyses estimated the comparative efficacy of ipilimumab and vemurafenib in the absence of head-to-head patient-level data for all trials and proportional hazards in overall survival.
Paoluzzi L, Hanniford D, Sokolova E, et al.
BET and BRAF inhibitors act synergistically against BRAF-mutant melanoma.
Cancer Med. 2016; 5(6):1183-93 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
BET and BRAF inhibitors act synergistically against BRAF-mutant melanoma.
Cancer Med. 2016; 5(6):1183-93 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Despite major advances in the treatment of metastatic melanoma, treatment failure is still inevitable in most cases. Manipulation of key epigenetic regulators, including inhibition of Bromodomain and extra-terminal domain (BET) family members impairs cell proliferation in vitro and tumor growth in vivo in different cancers, including melanoma. Here, we investigated the effect of combining the BET inhibitor JQ1 with the BRAF inhibitor Vemurafenib in in vitro and in vivo models of BRAF-mutant melanoma. We performed cytotoxicity and apoptosis assays, and a xenograft mouse model to determine the in vitro and in vivo efficacy of JQ1 in combination with Vemurafenib against BRAF-mutant melanoma cell lines. Further, to investigate the molecular mechanisms underlying the effects of combined treatment, we conducted antibody arrays of in vitro drug-treated cell lines and RNA sequencing of drug-treated xenograft tumors. The combination of JQ1 and Vemurafenib acted synergistically in BRAF-mutant cell lines, resulting in marked apoptosis in vitro, with upregulation of proapoptotic proteins. In vivo, combination treatment suppressed tumor growth and significantly improved survival compared to either drug alone. RNA sequencing of tumor tissues revealed almost four thousand genes that were uniquely modulated by the combination, with several anti-apoptotic genes significantly down-regulated. Collectively, our data provide a rationale for combined BET and BRAF inhibition as a novel strategy for the treatment of melanoma.
Stein J, Mann J
Specialty pharmacy services for patients receiving oral medications for solid tumors.
Am J Health Syst Pharm. 2016; 73(11):775-96 [PubMed] Related Publications
Specialty pharmacy services for patients receiving oral medications for solid tumors.
Am J Health Syst Pharm. 2016; 73(11):775-96 [PubMed] Related Publications
PURPOSE: Currently available oral oncology therapies are reviewed, and specialty pharmacy services for patients receiving these drugs are described.
SUMMARY: Market introductions of new oral oncology drugs have increased substantially over the past decade, and 25-30% of all oncology agents in development are oral medications. Oral agents for treatment of breast cancer include capecitabine, lafatinib, and palbociclib. Several oral agents are used in treating patients with lung cancer driven by mutations of genes coding for anaplastic lymphoma kinase (ALK) and epidermal growth factor receptor (EGFR); currently available agents include the ALK inhibitors certinib and crizotinib and the EGFR inhibitors afatinib, erlotinib, and gefitinib. Four oral targeted therapies are used in the treatment of melanoma associated with the B-Raf proto-oncogene, BRAF: cobimetinib, dabrafenib, trametinib, and vemurafenib. Oral agents for treatment of prostate cancer include abiraterone acetate and enzalutamide. Oral agents for treatment of renal cell carcinoma include axitinib, everolimus, pazopanib, sorafenib, and sunitinib. Specialty pharmacy services for patients receiving oral oncology agents can include (1) providing patient counseling and education on adverse effects and self-management strategies, (2) processing prior-authorization requests and helping patients navigate copayment assistance programs, and (3) monitoring for medication toxicities and recommending dose adjustments as appropriate.
CONCLUSION: Many oral oncology medications have been introduced over the past 10-15 years, with many others in clinical development. Due to the complexity of initiating and monitoring patients receiving these oral therapies, specialty pharmacy services are an essential component of many patients' cancer care.
SUMMARY: Market introductions of new oral oncology drugs have increased substantially over the past decade, and 25-30% of all oncology agents in development are oral medications. Oral agents for treatment of breast cancer include capecitabine, lafatinib, and palbociclib. Several oral agents are used in treating patients with lung cancer driven by mutations of genes coding for anaplastic lymphoma kinase (ALK) and epidermal growth factor receptor (EGFR); currently available agents include the ALK inhibitors certinib and crizotinib and the EGFR inhibitors afatinib, erlotinib, and gefitinib. Four oral targeted therapies are used in the treatment of melanoma associated with the B-Raf proto-oncogene, BRAF: cobimetinib, dabrafenib, trametinib, and vemurafenib. Oral agents for treatment of prostate cancer include abiraterone acetate and enzalutamide. Oral agents for treatment of renal cell carcinoma include axitinib, everolimus, pazopanib, sorafenib, and sunitinib. Specialty pharmacy services for patients receiving oral oncology agents can include (1) providing patient counseling and education on adverse effects and self-management strategies, (2) processing prior-authorization requests and helping patients navigate copayment assistance programs, and (3) monitoring for medication toxicities and recommending dose adjustments as appropriate.
CONCLUSION: Many oral oncology medications have been introduced over the past 10-15 years, with many others in clinical development. Due to the complexity of initiating and monitoring patients receiving these oral therapies, specialty pharmacy services are an essential component of many patients' cancer care.
Kaur A, Webster MR, Marchbank K, et al.
sFRP2 in the aged microenvironment drives melanoma metastasis and therapy resistance.
Nature. 2016; 532(7598):250-4 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
sFRP2 in the aged microenvironment drives melanoma metastasis and therapy resistance.
Nature. 2016; 532(7598):250-4 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Cancer is a disease of ageing. Clinically, aged cancer patients tend to have a poorer prognosis than young. This may be due to accumulated cellular damage, decreases in adaptive immunity, and chronic inflammation. However, the effects of the aged microenvironment on tumour progression have been largely unexplored. Since dermal fibroblasts can have profound impacts on melanoma progression, we examined whether age-related changes in dermal fibroblasts could drive melanoma metastasis and response to targeted therapy. Here we find that aged fibroblasts secrete a Wnt antagonist, sFRP2, which activates a multi-step signalling cascade in melanoma cells that results in a decrease in β-catenin and microphthalmia-associated transcription factor (MITF), and ultimately the loss of a key redox effector, APE1. Loss of APE1 attenuates the response of melanoma cells to DNA damage induced by reactive oxygen species, rendering the cells more resistant to targeted therapy (vemurafenib). Age-related increases in sFRP2 also augment both angiogenesis and metastasis of melanoma cells. These data provide an integrated view of how fibroblasts in the aged microenvironment contribute to tumour progression, offering new possibilities for the design of therapy for the elderly.
Spain L, Julve M, Larkin J
Combination dabrafenib and trametinib in the management of advanced melanoma with BRAFV600 mutations.
Expert Opin Pharmacother. 2016; 17(7):1031-8 [PubMed] Related Publications
Combination dabrafenib and trametinib in the management of advanced melanoma with BRAFV600 mutations.
Expert Opin Pharmacother. 2016; 17(7):1031-8 [PubMed] Related Publications
INTRODUCTION: In the 40-50% of advanced melanoma patients with tumors harboring BRAF V600E and V600 K mutations, BRAF inhibitors such as dabrafenib are a highly effective treatment. However, most patients develop resistance after several months on treatment. The addition of a MEK inhibitor, such as trametinib, to BRAF inhibition mitigates one key pathway of resistance, further increasing response rates and improving survival.
AREAS COVERED: This article summarizes the mechanism of action of the combination of dabrafenib and trametinib, its evolution through Phase I, II and III clinical trials and discusses its current use in the management of patients with advanced melanoma.
EXPERT OPINION: Combination therapy with dabrafenib and trametinib improves response rate, progression-free survival and overall survival when compared to dabrafenib or vemurafenib alone. The addition of trametinib to dabrafenib changes the adverse event profile, making hyperkeratosis and cutaneous squamous cell carcinomas less common but side effects such as fever and nausea more common. How dabrafenib/trametinib is best sequenced with other effective treatments such as immune checkpoint blockade remains uncertain.
AREAS COVERED: This article summarizes the mechanism of action of the combination of dabrafenib and trametinib, its evolution through Phase I, II and III clinical trials and discusses its current use in the management of patients with advanced melanoma.
EXPERT OPINION: Combination therapy with dabrafenib and trametinib improves response rate, progression-free survival and overall survival when compared to dabrafenib or vemurafenib alone. The addition of trametinib to dabrafenib changes the adverse event profile, making hyperkeratosis and cutaneous squamous cell carcinomas less common but side effects such as fever and nausea more common. How dabrafenib/trametinib is best sequenced with other effective treatments such as immune checkpoint blockade remains uncertain.
Boespflug A, Thomas L
Cobimetinib and vemurafenib for the treatment of melanoma.
Expert Opin Pharmacother. 2016; 17(7):1005-11 [PubMed] Related Publications
Cobimetinib and vemurafenib for the treatment of melanoma.
Expert Opin Pharmacother. 2016; 17(7):1005-11 [PubMed] Related Publications
INTRODUCTION: Cobimetinib combined with vemurafenib is a new approved MEK inhibitor for first line treatment of metastatic melanoma patients with BRAF V600 mutations. It improves tumor response rates and progression free survival compared to vemurafenib alone, while decreasing toxicities due to the paradoxical activation of the MAPK signaling pathway.
AREAS COVERED: This review covers the pharmacology, efficacy, and toxicity data derived from clinical and preclinical studies on cobimetinib. It also reports ongoing trials evaluating cobimetinib to better understand future developments for this drug.
EXPERT OPINION: The combination of cobimetinib and vemurafenib seems to be more toxic than the combination therapy dabrafenib and trametinib even if these four drugs have never been compared in a randomized trial. The future of this combination depends on its capacity to be combined simultaneously or sequentially with immune based therapies to improve the durability of responses.
AREAS COVERED: This review covers the pharmacology, efficacy, and toxicity data derived from clinical and preclinical studies on cobimetinib. It also reports ongoing trials evaluating cobimetinib to better understand future developments for this drug.
EXPERT OPINION: The combination of cobimetinib and vemurafenib seems to be more toxic than the combination therapy dabrafenib and trametinib even if these four drugs have never been compared in a randomized trial. The future of this combination depends on its capacity to be combined simultaneously or sequentially with immune based therapies to improve the durability of responses.
Foster SA, Whalen DM, Özen A, et al.
Activation Mechanism of Oncogenic Deletion Mutations in BRAF, EGFR, and HER2.
Cancer Cell. 2016; 29(4):477-93 [PubMed] Related Publications
Activation Mechanism of Oncogenic Deletion Mutations in BRAF, EGFR, and HER2.
Cancer Cell. 2016; 29(4):477-93 [PubMed] Related Publications
Activating mutations in protein kinases drive many cancers. While how recurring point mutations affect kinase activity has been described, the effect of in-frame deletions is not well understood. We show that oncogenic deletions within the β3-αC loop of HER2 and BRAF are analogous to the recurrent EGFR exon 19 deletions. We identify pancreatic carcinomas with BRAF deletions mutually exclusive with KRAS mutations. Crystal structures of BRAF deletions reveal the truncated loop restrains αC in an active "in" conformation, imparting resistance to inhibitors like vemurafenib that bind the αC "out" conformation. Characterization of loop length explains the prevalence of five amino acid deletions in BRAF, EGFR, and HER2 and highlights the importance of this region for kinase activity and inhibitor efficacy.
Keating GM
Cobimetinib Plus Vemurafenib: A Review in BRAF (V600) Mutation-Positive Unresectable or Metastatic Melanoma.
Drugs. 2016; 76(5):605-15 [PubMed] Related Publications
Cobimetinib Plus Vemurafenib: A Review in BRAF (V600) Mutation-Positive Unresectable or Metastatic Melanoma.
Drugs. 2016; 76(5):605-15 [PubMed] Related Publications
The MEK inhibitor cobimetinib (Cotellic(®)) is indicated for the treatment of patients with BRAF (V600) mutation-positive unresectable or metastatic melanoma, in combination with the BRAF inhibitor vemurafenib (Zelboraf(®)). In the pivotal coBRIM trial, previously untreated patients with BRAF (V600) mutation-positive unresectable, stage IIIC or stage IV melanoma received cobimetinib 60 mg once daily for the first 21 days of each 28-day cycle plus vemurafenib 960 mg twice daily or vemurafenib alone. Compared with vemurafenib alone, cobimetinib plus vemurafenib significantly prolonged progression-free survival (primary endpoint) and was associated with a significantly higher overall response rate and significantly prolonged overall survival. Cobimetinib plus vemurafenib had a manageable tolerability profile. In conclusion, cobimetinib plus vemurafenib is a valuable option for use in BRAF (V600) mutation-positive unresectable or metastatic melanoma.
Arance AM, Berrocal A, Lopez-Martin JA, et al.
Safety of vemurafenib in patients with BRAF (V600) mutated metastatic melanoma: the Spanish experience.
Clin Transl Oncol. 2016; 18(11):1147-1157 [PubMed] Related Publications
Safety of vemurafenib in patients with BRAF (V600) mutated metastatic melanoma: the Spanish experience.
Clin Transl Oncol. 2016; 18(11):1147-1157 [PubMed] Related Publications
OBJECTIVES: Vemurafenib tolerability was assessed in a large, open-label, multicentre study in patients with BRAF (V600) mutated advanced melanoma. We investigated safety, tolerability and efficacy of vemurafenib in Spanish patients participating in that study.
METHODS: Patients with previously treated or treatment-naive, unresectable stage IIIC or stage IV, BRAF (V600) mutation-positive melanoma received vemurafenib 960 mg twice daily until disease progression, unacceptable toxicity, withdrawal of consent or death. The primary endpoint was safety; secondary endpoints included overall response rate (ORR), progression-free survival (PFS) and overall survival (OS).
RESULTS: 301 Spanish patients were included, 70 % with M1c disease, 22 % with brain metastases and 51 % with prior systemic therapy for metastatic disease. Most frequent adverse events included fatigue (48 %), arthralgia (45 %), rash (41 %), photosensitivity (34 %) and skin neoplasms (21 %). Grade 3/4 adverse events occurred in 156 patients (52 %), including cutaneous squamous cell carcinoma (including keratoacanthoma; 16 %), fatigue (6 %) and arthralgia (5 %). The ORR was 28 % (95 % CI 23-34 %). Responses occurred in patients with brain metastases (18 %), elevated baseline lactate dehydrogenase (19 %) and poor performance status (15 %), and elderly patients (22 %). Median PFS was 5.8 (95 % CI 5.0-6.4) months; median OS was 10.5 (95 % CI 9.5-13.5) months.
CONCLUSION: Our results for Spanish patients in the vemurafenib safety study indicate similar efficacy and a comparable safety profile in Spanish patients with no new safety signals compared with the overall population. Clinical benefit was demonstrated in poor-prognosis patients and in those with favourable baseline characteristics, suggesting that poor-prognosis patients may also benefit from vemurafenib treatment.
METHODS: Patients with previously treated or treatment-naive, unresectable stage IIIC or stage IV, BRAF (V600) mutation-positive melanoma received vemurafenib 960 mg twice daily until disease progression, unacceptable toxicity, withdrawal of consent or death. The primary endpoint was safety; secondary endpoints included overall response rate (ORR), progression-free survival (PFS) and overall survival (OS).
RESULTS: 301 Spanish patients were included, 70 % with M1c disease, 22 % with brain metastases and 51 % with prior systemic therapy for metastatic disease. Most frequent adverse events included fatigue (48 %), arthralgia (45 %), rash (41 %), photosensitivity (34 %) and skin neoplasms (21 %). Grade 3/4 adverse events occurred in 156 patients (52 %), including cutaneous squamous cell carcinoma (including keratoacanthoma; 16 %), fatigue (6 %) and arthralgia (5 %). The ORR was 28 % (95 % CI 23-34 %). Responses occurred in patients with brain metastases (18 %), elevated baseline lactate dehydrogenase (19 %) and poor performance status (15 %), and elderly patients (22 %). Median PFS was 5.8 (95 % CI 5.0-6.4) months; median OS was 10.5 (95 % CI 9.5-13.5) months.
CONCLUSION: Our results for Spanish patients in the vemurafenib safety study indicate similar efficacy and a comparable safety profile in Spanish patients with no new safety signals compared with the overall population. Clinical benefit was demonstrated in poor-prognosis patients and in those with favourable baseline characteristics, suggesting that poor-prognosis patients may also benefit from vemurafenib treatment.
Tate SC, Burke TF, Hartman D, et al.
Optimising the combination dosing strategy of abemaciclib and vemurafenib in BRAF-mutated melanoma xenograft tumours.
Br J Cancer. 2016; 114(6):669-79 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Optimising the combination dosing strategy of abemaciclib and vemurafenib in BRAF-mutated melanoma xenograft tumours.
Br J Cancer. 2016; 114(6):669-79 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
BACKGROUND: Resistance to BRAF inhibition is a major cause of treatment failure for BRAF-mutated metastatic melanoma patients. Abemaciclib, a cyclin-dependent kinase 4 and 6 inhibitor, overcomes this resistance in xenograft tumours and offers a promising drug combination. The present work aims to characterise the quantitative pharmacology of the abemaciclib/vemurafenib combination using a semimechanistic pharmacokinetic/pharmacodynamic modelling approach and to identify an optimum dosing regimen for potential clinical evaluation.
METHODS: A PK/biomarker model was developed to connect abemaciclib/vemurafenib concentrations to changes in MAPK and cell cycle pathway biomarkers in A375 BRAF-mutated melanoma xenografts. Resultant tumour growth inhibition was described by relating (i) MAPK pathway inhibition to apoptosis, (ii) mitotic cell density to tumour growth and, under resistant conditions, (iii) retinoblastoma protein inhibition to cell survival.
RESULTS: The model successfully described vemurafenib/abemaciclib-mediated changes in MAPK pathway and cell cycle biomarkers. Initial tumour shrinkage by vemurafenib, acquisition of resistance and subsequent abemaciclib-mediated efficacy were successfully captured and externally validated. Model simulations illustrate the benefit of intermittent vemurafenib therapy over continuous treatment, and indicate that continuous abemaciclib in combination with intermittent vemurafenib offers the potential for considerable tumour regression.
CONCLUSIONS: The quantitative pharmacology of the abemaciclib/vemurafenib combination was successfully characterised and an optimised, clinically-relevant dosing strategy was identified.
METHODS: A PK/biomarker model was developed to connect abemaciclib/vemurafenib concentrations to changes in MAPK and cell cycle pathway biomarkers in A375 BRAF-mutated melanoma xenografts. Resultant tumour growth inhibition was described by relating (i) MAPK pathway inhibition to apoptosis, (ii) mitotic cell density to tumour growth and, under resistant conditions, (iii) retinoblastoma protein inhibition to cell survival.
RESULTS: The model successfully described vemurafenib/abemaciclib-mediated changes in MAPK pathway and cell cycle biomarkers. Initial tumour shrinkage by vemurafenib, acquisition of resistance and subsequent abemaciclib-mediated efficacy were successfully captured and externally validated. Model simulations illustrate the benefit of intermittent vemurafenib therapy over continuous treatment, and indicate that continuous abemaciclib in combination with intermittent vemurafenib offers the potential for considerable tumour regression.
CONCLUSIONS: The quantitative pharmacology of the abemaciclib/vemurafenib combination was successfully characterised and an optimised, clinically-relevant dosing strategy was identified.
Spagnolo F, Picasso V, Lambertini M, et al.
Survival of patients with metastatic melanoma and brain metastases in the era of MAP-kinase inhibitors and immunologic checkpoint blockade antibodies: A systematic review.
Cancer Treat Rev. 2016; 45:38-45 [PubMed] Related Publications
Survival of patients with metastatic melanoma and brain metastases in the era of MAP-kinase inhibitors and immunologic checkpoint blockade antibodies: A systematic review.
Cancer Treat Rev. 2016; 45:38-45 [PubMed] Related Publications
BACKGROUND: The incidence of brain metastases (BM) in melanoma patients is common and associated with poor prognosis. MAP-kinase inhibitors and immunologic checkpoint blockade antibodies led to improved survival of metastatic melanoma patients; however, patients with BM are under-represented or excluded from the majority of clinical trials and the impact of new drugs on their survival is less clear. With the present systematic review, we aimed to analyze outcomes of patients with melanoma BM treated with the new drugs, both in the setting of phase I-II-III clinical trials and in the "real world".
METHODS: An electronic search was performed to identify studies reporting survival outcomes of patients with melanoma BM treated with MAP-kinase inhibitors and/or immunologic checkpoint blockade antibodies, regardless of study design.
RESULTS: Twenty-two studies were included for a total of 2153 patients. Median OS was 7.9 months in phase I-II-III trials and 7.7 months in "real world" studies. In clinical trials, median OS was 7.0 months for patients treated with immunotherapy and 7.9 months for patients treated with BRAF inhibitors. In "real world" studies, median OS was 4.3 months and 7.7 months for patients treated with immunotherapy and BRAF inhibitors, respectively. Evidence of clinical activity exists for both immunotherapy and MAP-kinase inhibitors.
CONCLUSIONS: MAP-kinase inhibitors and immunologic checkpoint blockade antibodies have clinical activity and may achieve improved OS in patients with metastatic melanoma and BM. These results support the inclusion of patients with BM in investigations of new agents and new treatment regimens for metastatic melanoma.
METHODS: An electronic search was performed to identify studies reporting survival outcomes of patients with melanoma BM treated with MAP-kinase inhibitors and/or immunologic checkpoint blockade antibodies, regardless of study design.
RESULTS: Twenty-two studies were included for a total of 2153 patients. Median OS was 7.9 months in phase I-II-III trials and 7.7 months in "real world" studies. In clinical trials, median OS was 7.0 months for patients treated with immunotherapy and 7.9 months for patients treated with BRAF inhibitors. In "real world" studies, median OS was 4.3 months and 7.7 months for patients treated with immunotherapy and BRAF inhibitors, respectively. Evidence of clinical activity exists for both immunotherapy and MAP-kinase inhibitors.
CONCLUSIONS: MAP-kinase inhibitors and immunologic checkpoint blockade antibodies have clinical activity and may achieve improved OS in patients with metastatic melanoma and BM. These results support the inclusion of patients with BM in investigations of new agents and new treatment regimens for metastatic melanoma.
Atiq R, Hertz R, Eldad S, et al.
Suppression of B-Raf(V600E) cancers by MAPK hyper-activation.
Oncotarget. 2016; 7(14):18694-704 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Suppression of B-Raf(V600E) cancers by MAPK hyper-activation.
Oncotarget. 2016; 7(14):18694-704 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
B-Raf(V600E) activates MEK/MAPK signalling and acts as oncogenic driver of a variety of cancers, including melanoma, colorectal and papillary thyroid carcinoma. Specific B-Raf(V600E) kinase inhibitors (e.g., Vemurafenib) prove initial efficacy in melanoma followed shortly by acquired resistance, while failing in most other B-Raf(V600E) cancers due to primary resistance. Resistance is due to acquired mutations in the Ras/Raf/MEK/MAPK pathway and/or other oncogenic drivers that bypass B-Raf(V600E). Surprisingly, hyper-activation of MAPK by inhibiting its protein phosphatase 2A by a synthetic long-chain fatty acid analogue (MEDICA), results in oncogene-induced growth arrest and apoptosis of B-Raf(V600E) cancer cells. Growth arrest is accompanied by MAPK-mediated serine/threonine phosphorylation and suppression of a variety of oncogenic drivers that resist treatment by B-Raf(V600E) kinase inhibitors, including ErbB members, c-Met, IGFR, IRS, STAT3 and Akt. The combined activities of mutated B-Raf and MEDICA are required for generating hyper-activated MAPK, growth arrest and apoptosis, implying strict specificity for mutated B-Raf cancer cells.
Vázquez-Osorio I, Sánchez-Aguilar MD, García-Rodiño S, et al.
Vemurafenib-Induced Neutrophilic Panniculitis: A New Case and Review of the Literature.
Am J Dermatopathol. 2016; 38(7):e93-6 [PubMed] Related Publications
Vemurafenib-Induced Neutrophilic Panniculitis: A New Case and Review of the Literature.
Am J Dermatopathol. 2016; 38(7):e93-6 [PubMed] Related Publications
Vemurafenib has proved to be useful in the treatment of patients with unresectable or metastatic melanoma harboring the BRAF-V600E mutation, with better rates of overall and progression-free survival than previous treatments. Adverse cutaneous effects, such as alopecia, pruritus, photosensitivity reactions, verrucous keratosis, keratoacanthomas, or squamous cell carcinomas, have been described. Thirty cases of vemurafenib-associated panniculitis are available in the literature with variable clinical relevance. Only 9 of them exhibited definitive evidence of neutrophilic panniculitis. They all consist of multiple lesions, usually located in the lower limbs. Histopathologically, they have been described as predominantly neutrophilic, lymphocytic, or mixed, more commonly with lobular location. We report an additional case of neutrophilic panniculitis in a 45-year-old woman treated with vemurafenib for metastatic melanoma, presenting as a single lesion on his right leg. The lesion resolved spontaneously and did not need treatment reduction. The presentation of this condition with a single lesion is particularly challenging. Recognition of this association is important given the increasing use of vemurafenib and the potential implications of treatment withdrawal.
Hurabielle C, Pillebout E, Stehlé T, et al.
Mechanisms Underpinning Increased Plasma Creatinine Levels in Patients Receiving Vemurafenib for Advanced Melanoma.
PLoS One. 2016; 11(3):e0149873 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Mechanisms Underpinning Increased Plasma Creatinine Levels in Patients Receiving Vemurafenib for Advanced Melanoma.
PLoS One. 2016; 11(3):e0149873 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
CONTEXT: Serum creatinine has been reported to increase in patients receiving Vemurafenib, yet neither the prevalence nor the mechanism of this adverse event are known.
OBJECTIVE: We aimed to evaluate the frequency and the mechanisms of increases in plasma creatinine level in patients receiving Vemurafenib for advanced melanoma.
METHODS: We performed a retrospective monocentric study including consecutive patients treated with Vemurafenib for an advanced melanoma. We collected clinical and biological data concerning renal function before introduction of Vemurafenib and in the course of monthly follow-up visits from March 2013 to December 2014. Cystatin C-derived glomerular filtration rate was evaluated before and after Vemurafenib initiation, as increase in serum cystatin C is specific to a decrease in the glomerular filtration rate. We also performed thorough renal explorations in 3 patients, with measurement of tubular secretion of creatinine before and after Vemurafenib initiation and a renal biopsy in 2 patients.
RESULTS: 70 patients were included: 97% of them displayed an immediate, and thereafter stable, increase in creatinine (+22.8%) after Vemurafenib initiation. In 44/52 patients in whom Vemurafenib was discontinued, creatinine levels returned to baseline. Serum cystatin C increased, although proportionally less than serum creatinine, showing that creatinine increase under vemurafenib was indeed partly due to a renal function impairment. In addition, renal explorations demonstrated that Vemurafenib induced an inhibition of creatinine tubular secretion.
CONCLUSION: Thus, Vemurafenib induces a dual mechanism of increase in plasma creatinine with both an inhibition of creatinine tubular secretion and slight renal function impairment. However, this side effect is mostly reversible when Vemurafenib is discontinued, and should not lead physicians to discontinue the treatment if it is effective.
OBJECTIVE: We aimed to evaluate the frequency and the mechanisms of increases in plasma creatinine level in patients receiving Vemurafenib for advanced melanoma.
METHODS: We performed a retrospective monocentric study including consecutive patients treated with Vemurafenib for an advanced melanoma. We collected clinical and biological data concerning renal function before introduction of Vemurafenib and in the course of monthly follow-up visits from March 2013 to December 2014. Cystatin C-derived glomerular filtration rate was evaluated before and after Vemurafenib initiation, as increase in serum cystatin C is specific to a decrease in the glomerular filtration rate. We also performed thorough renal explorations in 3 patients, with measurement of tubular secretion of creatinine before and after Vemurafenib initiation and a renal biopsy in 2 patients.
RESULTS: 70 patients were included: 97% of them displayed an immediate, and thereafter stable, increase in creatinine (+22.8%) after Vemurafenib initiation. In 44/52 patients in whom Vemurafenib was discontinued, creatinine levels returned to baseline. Serum cystatin C increased, although proportionally less than serum creatinine, showing that creatinine increase under vemurafenib was indeed partly due to a renal function impairment. In addition, renal explorations demonstrated that Vemurafenib induced an inhibition of creatinine tubular secretion.
CONCLUSION: Thus, Vemurafenib induces a dual mechanism of increase in plasma creatinine with both an inhibition of creatinine tubular secretion and slight renal function impairment. However, this side effect is mostly reversible when Vemurafenib is discontinued, and should not lead physicians to discontinue the treatment if it is effective.
Waizenegger IC, Baum A, Steurer S, et al.
A Novel RAF Kinase Inhibitor with DFG-Out-Binding Mode: High Efficacy in BRAF-Mutant Tumor Xenograft Models in the Absence of Normal Tissue Hyperproliferation.
Mol Cancer Ther. 2016; 15(3):354-65 [PubMed] Related Publications
A Novel RAF Kinase Inhibitor with DFG-Out-Binding Mode: High Efficacy in BRAF-Mutant Tumor Xenograft Models in the Absence of Normal Tissue Hyperproliferation.
Mol Cancer Ther. 2016; 15(3):354-65 [PubMed] Related Publications
BI 882370 is a highly potent and selective RAF inhibitor that binds to the DFG-out (inactive) conformation of the BRAF kinase. The compound inhibited proliferation of human BRAF-mutant melanoma cells with 100× higher potency (1-10 nmol/L) than vemurafenib, whereas wild-type cells were not affected at 1,000 nmol/L. BI 882370 administered orally was efficacious in multiple mouse models of BRAF-mutant melanomas and colorectal carcinomas, and at 25 mg/kg twice daily showed superior efficacy compared with vemurafenib, dabrafenib, or trametinib (dosed to provide exposures reached in patients). To model drug resistance, A375 melanoma-bearing mice were initially treated with vemurafenib; all tumors responded with regression, but the majority subsequently resumed growth. Trametinib did not show any efficacy in this progressing population. BI 882370 induced tumor regression; however, resistance developed within 3 weeks. BI 882370 in combination with trametinib resulted in more pronounced regressions, and resistance was not observed during 5 weeks of second-line therapy. Importantly, mice treated with BI 882370 did not show any body weight loss or clinical signs of intolerability, and no pathologic changes were observed in several major organs investigated, including skin. Furthermore, a pilot study in rats (up to 60 mg/kg daily for 2 weeks) indicated lack of toxicity in terms of clinical chemistry, hematology, pathology, and toxicogenomics. Our results indicate the feasibility of developing novel compounds that provide an improved therapeutic window compared with first-generation BRAF inhibitors, resulting in more pronounced and long-lasting pathway suppression and thus improved efficacy.
Dummer R, Siano M, Hunger RE, et al.
The updated Swiss guidelines 2016 for the treatment and follow-up of cutaneous melanoma.
Swiss Med Wkly. 2016; 146:w14279 [PubMed] Related Publications
The updated Swiss guidelines 2016 for the treatment and follow-up of cutaneous melanoma.
Swiss Med Wkly. 2016; 146:w14279 [PubMed] Related Publications
Cutaneous melanoma is the most deadly cutaneous neoplasm. In order to guide treatment decisions and follow-up of melanoma patients, guidelines for the management of melanoma in Switzerland were inaugurated in 2001 and revised in 2006 and 2016. Recent data on surgical and medical treatments from randomised trials necessitated modification of the treatment and follow-up recommendations.
Holzer K, Drucker E, Oliver S, et al.
Cellular apoptosis susceptibility (CAS) is overexpressed in thyroid carcinoma and maintains tumor cell growth: A potential link to the BRAFV600E mutation.
Int J Oncol. 2016; 48(4):1679-87 [PubMed] Related Publications
Cellular apoptosis susceptibility (CAS) is overexpressed in thyroid carcinoma and maintains tumor cell growth: A potential link to the BRAFV600E mutation.
Int J Oncol. 2016; 48(4):1679-87 [PubMed] Related Publications
Thyroid carcinoma is among the most common malignant endocrine neoplasms with a rising incidence. Genetic alterations occurring in thyroid cancer frequently affect the RAS/RAF/MEK/ERK-pathway such as the oncogenic, kinase-activating BRAF(V600E) mutation. Nuclear transport receptors including importins and exportins represent an important part of the nuclear transport machinery providing nucleo-cytoplasmic exchange of macromolecules. The role of nuclear transport receptors in the development and progression of thyroid carcinomas is largely unknown. Here, we studied the expression and function of the exportin cellular apoptosis susceptibility (CAS) in thyroid carcinogenesis and its link to the BRAF(V600E) mutation. By using immunohistochemistry (IHC) we found significantly increased IHC scores of CAS in primary papillary (PTC) and medullary (MTC), but not in follicular (FTC) thyroid carcinoma compared to non-tumorous (NT) thyroid tissue. Interestingly, metastases of the aforementioned subtypes including FTC showed a strong CAS positivity. Among PTCs we observed that CAS immunoreactivity was significantly higher in the tumors harboring the BRAF(V600E) mutation. Furthermore, depletion of CAS by RNAi in the BRAF(V600E)-positive PTC cell line B-CPAP led to reduced tumor cell growth measured by crystal violet assays. This phenotype could be attributed to reduced proliferation and increased cell death as assayed by BrdU ELISAs and immunoblotting for PARP-cleavage, respectively. Finally, we found additive effects of CAS siRNA and vemurafenib treatment in B-CPAP cells. Collectively, these data suggest that CAS overexpression in thyroid carcinoma depends on the subtype and the disease stage. Our findings also indicate that CAS maintains PTC cell proliferation and survival. Targeting CAS could represent a potential therapeutic approach particularly in combination with BRAF inhibitors such as vemurafenib in BRAF(V600E)-positive tumors.
Tan J
Perianal melanoma with a BRAF gene mutation in a young Portuguese Roma native.
BMJ Case Rep. 2016; 2016 [PubMed] Related Publications
Perianal melanoma with a BRAF gene mutation in a young Portuguese Roma native.
BMJ Case Rep. 2016; 2016 [PubMed] Related Publications
A case of a young man diagnosed with perianal nodular melanoma with a gene mutation, accompanied by regional and pulmonary metastases on initial presentation, and later on with hepatic and bone involvement, is presented. The patient underwent wide local excision but was unresponsive to dacarbazine. Targeted therapy with vemurafenib had shown clinical improvement for a 5-month duration until he showed signs of disease progression. Just after the shift of adjuvant therapy to ipilimumab, he was diagnosed with multiple cerebral metastases that eventually led to his demise 6 months after initiation of vemurafenib, having had a 12-month survival period from the time of initial melanoma diagnosis.
Pich C, Teiti I, Sarrabayrouse G, et al.
Melanoma Expressed-CD70 Is Regulated by RhoA and MAPK Pathways without Affecting Vemurafenib Treatment Activity.
PLoS One. 2016; 11(2):e0148095 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
Melanoma Expressed-CD70 Is Regulated by RhoA and MAPK Pathways without Affecting Vemurafenib Treatment Activity.
PLoS One. 2016; 11(2):e0148095 [PubMed] Article available free on PMC after 15/03/2017 Related Publications
CD70 is a costimulatory molecule member of the Tumor Necrosis Factor family that is expressed on activated immune cells. Its ectopic expression has been described in several types of cancer cells including lymphomas, renal cell carcinomas and glioblastomas. We have recently described its expression in a part of tumor cells from the vast majority of melanoma biopsies and human melanoma cell lines, and found that CD70 expression decreased over time as the disease progressed. Here, we show that RhoA, BRAF and Mitogen Activating Protein Kinase pathways are involved in the positive transcriptional regulation of CD70 expression in melanomas. Interestingly, the clinical inhibitor of the common BRAF V600E/D variants, Vemurafenib (PLX-4032), which is currently used to treat melanoma patients with BRAF V600E/D-mutated metastatic melanomas, decreased CD70 expression in human CD70+ melanoma cell lines. This decrease was seen in melanoma cells both with and without the BRAFV600E/D mutation, although was less efficient in those lacking the mutation. But interestingly, by silencing CD70 in CD70+ melanoma cell lines we show that PLX-4032-induced melanoma cell killing and its inhibitory effect on MAPK pathway activation are unaffected by CD70 expression. Consequently, our work demonstrates that CD70 ectopic expression in melanomas is not a valuable biomarker to predict tumor cells sensitivity to BRAF V600 inhibitors.

 Monoclonal Antibodies
Monoclonal Antibodies