| Osteosarcoma |
|
|
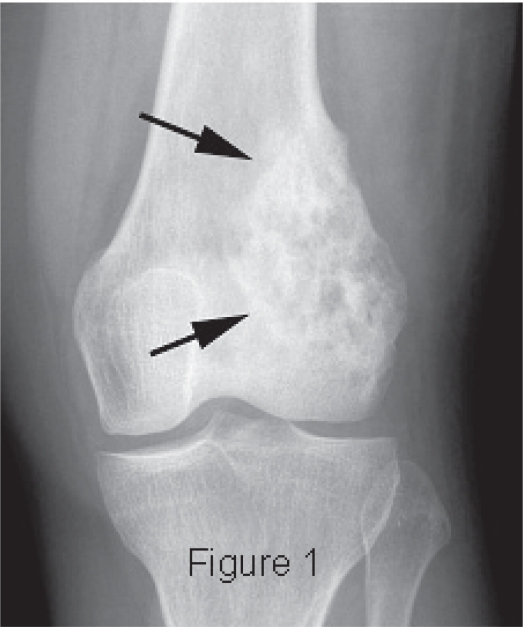 |
Osteogenic Sarcoma (osteosarcoma) is a bone forming cancer. It is the most frequent type of bone tumour and is most common between the ages of 15 to 25. Over 90% of tumours are located in the metaphysis (the growing ends of the bone), the most common sites are the bones around the knee which account for 80% of cases. Osteosarcomas vary greatly in radiological and pathological features and therefore needs careful diagnosis to differentiate this from other bone tumours. Most are high grade intramedullary osteosarcomas, about 5% are low grade lesions, some are secondary osteosarcomas (for example those caused by radiation therapy).
Figure 1. Radiograph showing an osteolytic and osteoblastic intra-medullary tumor characteristic of osteosarcoma. From Layfield J et al. Clin Med Pathol. 2008; 1: 55-59. Available under a Creative Commons CC-BY-3.0 license. |
Menu: Osteosarcoma
 Information for Patients and Family
Information for Patients and Family Information for Health Professionals / Researchers
Information for Health Professionals / Researchers Latest Research Publications
Latest Research Publications Osteosarcoma FAQs
Osteosarcoma FAQs Bone Cancer Resources
Bone Cancer ResourcesInformation Patients and Family (13 links)
 Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
National Cancer Institute![]() PDQ summaries are written and frequently updated by editorial boards of experts Further info.
PDQ summaries are written and frequently updated by editorial boards of experts Further info.
NHS Choices![]() NHS Choices information is quality assured by experts and content is reviewed at least every 2 years. Further info.
NHS Choices information is quality assured by experts and content is reviewed at least every 2 years. Further info.
Overview of promary bone cancers in general, though does include some specific information about Osteosarcoma, Ewing's Sarcoma, Chondrosarcoma and Spindle cell sarcoma. Ewing's Sarcoma Ewing's Sarcoma Ewing's Sarcoma - Molecular Biology
![]() Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
BCRT became a registered the charity in 2006 and raises funds for research into primary bone cancer, and provides information and support for patients and their families. The Website includes information booklets, personal stories and a section for teenagers. Bone Cancers Ewing's Sarcoma Ewing's Sarcoma Ewing's Sarcoma - Molecular Biology
Macmillan Cancer Support![]() Content is developed by a team of information development nurses and content editors, and reviewed by health professionals. Further info.
Content is developed by a team of information development nurses and content editors, and reviewed by health professionals. Further info.
Cancer.Net![]() Content is peer reviewed and Cancer.Net has an Editorial Board of experts and advocates. Content is reviewed annually or as needed. Further info.
Content is peer reviewed and Cancer.Net has an Editorial Board of experts and advocates. Content is reviewed annually or as needed. Further info.
Bone Cancer Research Trust![]() Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Information is reviewed by a panel of scientific and clinical experts, patients, parents/ carers, Further info.
Overview of Osteosarcoma and the different types of the disease.
 Osteosarcoma
Osteosarcoma
Mayo Clinic
Dr. Carola Arndt discusses osteosarcoma which is one of the most common malignant tumors of bone in teenagers and young adults. She discusses diagnosis, evaluation, and treatment of osteosarcoma. Also mentions the Children's Oncology group and EURAMOS study. Mentions the multi-disciplinary approach at Mayo Clinic.
 European and American Osteosarcoma Study Group
European and American Osteosarcoma Study Group
EURAMOS
Includes information about the EURAMOS-1 clinical trial. There is also detailed information about osteosarcoma and patients' art work.
 Liddy Shriver Sarcoma Initiative
Liddy Shriver Sarcoma Initiative
Founded in 2003 the initiative aims improve the quality of life for people dealing with sarcomas around the world, raising awareness and research funds. It has an international panel of medical experts. Ewing's Sarcoma Soft Tissue Sarcomas Ewing's Sarcoma Ewing's Sarcoma - Molecular Biology
American Cancer Society
A detailed guides
Childrens' Oncology Group
Includes information, with sections on newly diagnosed, in treatment and after treatment.
A charity for bone and soft-tissue sarcomas in the UK. The organisation raises funds for research and provides information and support.
 Sarcoma UK Online Support Group
Sarcoma UK Online Support Group
Sarcoma UK
This group is for the benefit of sarcoma patients, carers and their families being treated in the UK on the NHS or privately.
Information for Health Professionals / Researchers (9 links)
- PubMed search for publications about Osteosarcoma - Limit search to: [Reviews]
PubMed Central search for free-access publications about Osteosarcoma
MeSH term: Osteosarcoma US National Library of Medicine
US National Library of Medicine PubMed has over 22 million citations for biomedical literature from MEDLINE, life science journals, and online books. Constantly updated.
PubMed has over 22 million citations for biomedical literature from MEDLINE, life science journals, and online books. Constantly updated.
 Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
Osteosarcoma and Malignant Fibrous Histiocytoma of Bone Treatment
National Cancer Institute![]() PDQ summaries are written and frequently updated by editorial boards of experts Further info.
PDQ summaries are written and frequently updated by editorial boards of experts Further info.
A website by orthopedic surgeon Dr. Henry DeGroot, with contributions from numerous clinical colleagues. It includes numerous case studies, including radiology and pathology images, and information covering a comprehensive range of bone tumours. Bone Cancers Ewing's Sarcoma Ewing's Sarcoma Ewing's Sarcoma - Molecular Biology
 Case 10: A 12 year old male with an osteolytic lesion of the proximal left tibia
Case 10: A 12 year old male with an osteolytic lesion of the proximal left tibia
Department of Pathology, University of Pittsburgh
Department of Pathology, University of Pittsburgh
 Clinical Case: 51 year old with knee pain (small cell osteosarcoma)
Clinical Case: 51 year old with knee pain (small cell osteosarcoma)
BoneTumour.org
 European and American Osteosarcoma Study Group
European and American Osteosarcoma Study Group
EURAMOS
Includes information about the EURAMOS-1 clinical trial. There is also detailed information about osteosarcoma and patients' art work.
Medscape
A detailed clinical case covering an overview, workup/investigations and treatment and follow-up. The case includes radiology images.
Medscape
A detailed clinical case covering an overview, workup/investigations and treatment. The case includes radiology and pathology images.
 Genetic features of Osteosarcoma
Genetic features of OsteosarcomaLatest Research Publications
This list of publications is regularly updated (Source: PubMed).
Sacral Reconstruction with a 3D-Printed Implant after Hemisacrectomy in a Patient with Sacral Osteosarcoma: 1-Year Follow-Up Result.
Yonsei Med J. 2017; 58(2):453-457 [PubMed] Free Access to Full Article Related Publications
Related:
 Bone Cancers
Bone Cancers A predictive model to estimate the pretest probability of metastasis in patients with osteosarcoma.
Medicine (Baltimore). 2017; 96(3):e5909 [PubMed] Free Access to Full Article Related Publications
MiR-451 suppresses proliferation, migration and promotes apoptosis of the human osteosarcoma by targeting macrophage migration inhibitory factor.
Biomed Pharmacother. 2017; 87:621-627 [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  MicroRNAs
MicroRNAs  Angiogenesis and Cancer
Angiogenesis and Cancer  MIF
MIF Overexpression of miR-92a promotes the tumor growth of osteosarcoma by suppressing F-box and WD repeat-containing protein 7.
Gene. 2017; 606:10-16 [PubMed] Related Publications
Related:
 MicroRNAs
MicroRNAs  FBXW7
FBXW7 ViscumTT induces apoptosis and alters IAP expression in osteosarcoma in vitro and has synergistic action when combined with different chemotherapeutic drugs.
BMC Complement Altern Med. 2017; 17(1):26 [PubMed] Free Access to Full Article Related Publications
METHODS: Two osteosarcoma cell lines were treated with three different mistletoe extracts viscum, TT and viscumTT to compare their apoptotic potential. For this purpose, annexin/PI staining and caspase-3, -8 and -9 activity were investigated by flow cytometry. To determine the mechanism of action, alterations in expression of inhibitors of apoptosis (IAPs) were detected by western blot. Apoptosis induction by co-treatment of viscum, TT and viscumTT with doxorubicin, etoposide and ifosfamide was examined by flow cytometry.
RESULTS: In vitro as well as ex vivo, the whole mistletoe extract viscumTT led to strong inhibition of proliferation and synergistic apoptosis induction in osteosarcoma cells. In the investigations of mechanism of action, inhibitors of apoptosis such as XIAP, BIRC5 and CLSPN showed a clear down-regulation after viscumTT treatment. In addition, co-treatment with doxorubicin, etoposide and ifosfamide further enhanced apoptosis induction, also synergistically.
CONCLUSION: ViscumTT treatment results in synergistic apoptosis induction in osteosarcoma cells in vitro and ex vivo. Additionally, conventional standard chemotherapeutic drugs such as doxorubicin, etoposide and ifosfamide were able to dramatically enhance apoptosis induction. These results promise a high potential of viscumTT as an additional adjuvant therapy approach for osteosarcoma.
Related:
 Apoptosis
Apoptosis  Doxorubicin
Doxorubicin  Etoposide
Etoposide  BIRC5
BIRC5 AMD3100 combined with triptolide inhibit proliferation, invasion and metastasis and induce apoptosis of human U2OS osteosarcoma cells.
Biomed Pharmacother. 2017; 86:677-685 [PubMed] Related Publications
OBJECTIVE: This work aimed to investigate the effects of AMD3100 plus triptolide on the proliferation, apoptosis, invasion and metastasis of OS cells.
METHODS: The expression levels of SDF-1 and CXCR4 in five OS cell lines was analyzed by qRT-PCR, western blotting and ELISA assays. The effect of AMD3100 and triptolide on the proliferation, apoptosis and invasion of U2OS cells was evaluated by CCK-8, flow cytometry and transwell assay, respectively. Orthotopic intra-tibial growth and lung metastasis mouse model of OS were employed to evaluate the inhibition effect of AMD3100 and triptolide on primary OS growth and lung metastasis.
RESULTS: CXCR4 protein expression was detected in HOS-8603, MG-63, U2OS and 143B but not Saos2 cells, and all these cell lines expressed SDF-1. AMD3100 plus triptolide induced proliferation inhibition and apoptosis of U2OS cells, which was attributed to the downregulation of c-Myc, survivin, cyclin D1 and increased cleaved caspase-3 and PARP. AMD3100 and triptolide also suppressed SDF-1 induced invasion of CXCR4+ U2OS cells, which was validated by decreased expression of MMP-2 and 9, VEGF, m-Calpain and β-catenin. Moreover, the phosphorylation levels of Erk1/2, Akt and STAT3, as well as the nuclear translocation and phosphorylation of NF-κB p65 in U2OS cells were also reduced by AMD3100 and triptolide. In vivo, AMD3100 and triptolide significantly reduced primary tumor growth and lung metastasis of U2OS cells.
CONCLUSIONS: AMD3100 combined with triptolide can reduce proliferation and metastasis, and induce apoptosis of U2OS cells, which may be related to the Erk1/2, Akt, STAT3 and NF-κB pathways.
Related:
 Apoptosis
Apoptosis  Signal Transduction
Signal Transduction MicroRNA-195-5p suppresses osteosarcoma cell proliferation and invasion by suppressing naked cuticle homolog 1.
Cell Biol Int. 2017; 41(3):287-295 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers  MicroRNAs
MicroRNAs MicroRNA-210 is increased and it is required for dedifferentiation of osteosarcoma cell line.
Cell Biol Int. 2017; 41(3):267-275 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers  MicroRNAs
MicroRNAs Long Noncoding RNA PANDA Positively Regulates Proliferation of Osteosarcoma Cells.
Anticancer Res. 2017; 37(1):81-85 [PubMed] Related Publications
MATERIALS AND METHODS: U2OS cells were transfected with siRNAs against PANDA 72 h later and they were subjected to reverse transcription-polymerase chain reaction (RT-PCR), quantitative RT-PCR and cell-cycle analysis.
RESULTS: PANDA was highly expressed in U2OS cells, and its expression was induced by DNA damage. Silencing PANDA caused arrest at the G1 phase of the cell cycle, leading to inhibition of cell proliferation. Quantitative RT-PCR showed that silencing PANDA increased mRNA levels of the cyclin-dependent kinase inhibitor p18, which caused G1 phase arrest.
CONCLUSION: These results suggest that PANDA promotes G1-S transition by repressing p18 transcription, and thus promotes U2OS cell proliferation.
Related:
 Bone Cancers
Bone Cancers  Signal Transduction
Signal Transduction  CDKN2C
CDKN2C Correlation between TRAIL and caspase-8 expression and their relationship with cell proliferation and apoptosis in human osteosarcoma.
Genet Mol Res. 2016; 15(4) [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  TNFSF10
TNFSF10 The role of the IGF-1 Ec in myoskeletal system and osteosarcoma pathophysiology.
Crit Rev Oncol Hematol. 2016; 108:137-145 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers  IGF1
IGF1 IKKα inibition by a glucosamine derivative enhances Maspin expression in osteosarcoma cell line.
Chem Biol Interact. 2017; 262:19-28 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers  MMP9: matrix metallopeptidase 9
MMP9: matrix metallopeptidase 9  SERPINB5
SERPINB5 Targeted osteosarcoma chemotherapy using RGD peptide-installed doxorubicin-loaded biodegradable polymeric micelle.
Biomed Pharmacother. 2017; 85:160-168 [PubMed] Related Publications
Related:
 Doxorubicin
Doxorubicin Long Noncoding RNA EWSAT1 Promotes Osteosarcoma Cell Growth and Metastasis Through Suppression of MEG3 Expression.
DNA Cell Biol. 2016; 35(12):812-818 [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  Signal Transduction
Signal Transduction Parthenolide Induces Reactive Oxygen Species-Mediated Autophagic Cell Death in Human Osteosarcoma Cells.
Cell Physiol Biochem. 2016; 40(1-2):146-154 [PubMed] Related Publications
METHODS: In this study, the effects of parthenolide were evaluated and characterized in human osteosarcoma cancer cell. Cell viability was assessed by CCK-8. Apoptosis was assessed by Annexin V-FITC/PI Flow cytometry assay. Relative quantitative real-time PCR and western blot were used to determine the expressions of genes and proteins.
RESULTS: Our results suggest that parthenolide did not cause caspase-dependent cell death in osteosarcoma cancer cells, as indicated by the absence of significant early apoptosis as well as caspase-3 cleavage. Instead, parthenolide increased the autophagy and mitophagy, as characterized by increased PINK1 and Parkin translocation to mitochondria and enhanced autophagy proteins. The induction of autophagy by parthenolide was associated with the increase of reactive oxygen species (ROS). ROS antioxidants N-acetylcysteine (NAC) attenuated parthenolide-induced autophagy activity.
CONCLUSIONS: Our findings unveil a novel mechanism of drug action by parthenolide in osteosarcoma cancer cells and suggest a potential value of treating osteosarcoma cancer through a caspase-independent autophagic cell death by ROS activation.
Related:
 CASP3
CASP3 Oncogenic Properties of Candidate Oncogenes in Chromosome Region 17p11.2p12 in Human Osteosarcoma.
Cytogenet Genome Res. 2016; 150(1):52-59 [PubMed] Related Publications
Related:
 Chromosome 17
Chromosome 17  TOP1
TOP1  PMP22
PMP22 Clinical Outcome and Survival of Osteosarcoma Patients in Cipto Mangunkusumo Hospital: Limb Salvage Surgery versus Amputation.
Acta Med Indones. 2016; 48(3):175-183 [PubMed] Related Publications
METHODS: this is a retrospective cohort study of osteosarcoma patients in Cipto Mangunkusumo Hospital underwent limb salvage surgery (LSS), amputation, LSS + amputation, and refused surgery from year 1995 to 2014. The surgical decision was based on patient's age, staging, location, neurovascular involvement, Huvos type, functional demand, patient preference, and general condition. Functional outcome was assessed using the Musculoskeletal Tumor Society (MSTS) score with the maximum score of 30.
RESULTS: subjects consisted of 80 male and 52 female aged 4 to 61 year-old. They underwent limb salvage surgery (LSS) (n=37), amputation (n=42), LSS + amputation (n=2), and refused surgery (n=51). Overall 5-year cumulative survival rate was 14.6%. The 5-year survival rate for each group; LSS, amputation, combined LSS and amputation, and refused surgery was 34.8%; 15.9%; 0%; and 0%, respectively. Patients with tumor size <8 cm tend to underwent LSS compared to amputations (60.7% vs 39.3%, p=0.046). Local recurrence-free survival for LSS and amputation was 96.2% and 86.5% respectively (p=0.586). MSTS score was higher in LSS than amputation group (25.0 vs 18.5, p=0.011).
CONCLUSION: LSS had higher survival rate than amputation in osteosarcoma patients who were treated in Cipto Mangunkusumo Hospital. MSTS functional score in the LSS group was higher than amputation group.
Related:
 Bone Cancers
Bone Cancers Effects of resection margins on local recurrence of osteosarcoma in extremity and pelvis: Systematic review and meta-analysis.
Int J Surg. 2016; 36(Pt A):283-292 [PubMed] Related Publications
METHODS: EMBASE, PubMed and Cochrane CENTRAL were searched from January 1980 to July 2016. The quality of included studies was evaluated using the Newcastle-Ottawa Quality Assessment Scale. The odds ratio and 95% confidence interval of local recurrence were estimated, respectively, for inadequate vs adequate margins and marginal vs wide margins using a random-effect model. Chi-square test was performed to comparing the local recurrence rate between extremity and pelvic osteosarcomas with an identical surgical margin.
RESULTS: Thirteen articles involving 1559 patients (175 with and 1384 without local recurrence) were included in this study. The meta-analysis showed that the osteosarcoma resected with inadequate and marginal margins, whether in extremity or in pelvis, were associated with a significantly higher local recurrence rate than the osteosarcoma resected with adequate and wide margins, respectively. Chi-square test showed that, when pelvic and extremity osteosarcomas were removed with an identical resection margin, the local recurrence was significantly more frequent in pelvis osteosarcoma than in extremity osteosarcoma.
CONCLUSION: This study provides level IIa evidence to support that the surgery with adequate or wide resection margin has positive effect on reducing the risk of local recurrence in osteosarcoma. In addition, the factors independent of resection margin are more likely to increase the risk of local recurrence in pelvic osteosarcoma.
LEVEL OF EVIDENCE: Level IIa, Therapeutic study.
Related:
 Bone Cancers
Bone Cancers A 20-year retrospective analysis of CT-based pre-operative identification of pulmonary metastases in patients with osteosarcoma: A single-center review.
J Pediatr Surg. 2017; 52(1):115-119 [PubMed] Related Publications
METHODS: We reviewed OS thoracotomies performed at our institution from 1996 to 2015. The number of metastases identified on pre-operative chest CT was compared to the number of metastases seen on pathology (both metastases with viable cells and non-viable, osteoid-only metastases).
RESULTS: Eighty-eight patients underwent 161 thoracotomies with a median of 14days (range, 1-85) between CT and surgery, a median of 2 CT-identified lesions (range, 0-15), and a median of 4 resected lesions (range, 1-25). In 56 (34.8%) cases, more metastases were found surgically than were seen on CT, and among these, 34 (21.1%) had a greater number of viable metastases. There was poor overall correlation between CT and pathology findings (Kendall Tau-b=0.506), regardless of CT slice thickness, decade of thoracotomy, or total number of CT-identified lesions.
CONCLUSIONS: CT accuracy in pre-operatively quantifying OS pulmonary metastases has not improved in recent decades. Consequently, we recommend an open technique with direct lung palpation for complete identification and resection of OS pulmonary metastases.
LEVEL OF EVIDENCE: Level IV, retrospective study with no comparison group.
Related:
 Bone Cancers
Bone Cancers Inhibition of ERK1/2 Signaling Pathway is Involved in Melatonin's Antiproliferative Effect on Human MG-63 Osteosarcoma Cells.
Cell Physiol Biochem. 2016; 39(6):2297-2307 [PubMed] Related Publications
AIMS: The present study investigated whether MAPK and Akt signaling pathways are involved in melatonin's antiproliferative actions on the human MG-63 osteosarcoma cells.
METHODS/RESULTS: Western blot analysis confirmed that melatonin significantly inhibited phosphorylation of ERK1/2 but not p38, JNK, or Akt. The expression of ERK1/2, p38, JNK, and Akt was not altered by melatonin. PD98059 and melatonin alone, and especially in combination, significantly inhibited cell proliferation. The changes included G1 and G2/M phase arrest of the cell cycle, and a downregulation of the expression at both the protein and mRNA levels of cyclin D1 and CDK4 (related to the G1 phase) and of cyclin B1 and CDK1 (related to the G2/M phase) as measured by flow cytometry after propidium iodide staining, and both western blot and real-time PCR, respectively. Furthermore, the combination of PD98059 and melatonin synergistically and markedly augmented the action of either agent alone. Co-immunoprecipitation further confirmed that there was an interaction between p-ERK1/2 and cyclin D1, CDK4, cyclin B1, or CDK1, which was blunted in the presence of melatonin or PD98059.
CONCLUSION: These findings suggest that melatonin's antiproliferative action is mediated by inhibition of the ERK1/2 signaling pathway rather than the p38, JNK, or Akt pathways.
Related:
 AKT1
AKT1  BCL1 Gene (CCND1)
BCL1 Gene (CCND1) Co-delivery of doxorubicin and recombinant plasmid pHSP70-Plk1-shRNA by bacterial magnetosomes for osteosarcoma therapy.
Int J Nanomedicine. 2016; 11:5277-5286 [PubMed] Free Access to Full Article Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  Doxorubicin
Doxorubicin Osteosarcoma of pelvic bones: imaging features.
Clin Imaging. 2017 Jan - Feb; 41:59-64 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers Phyllanthus urinaria's Inhibition of Human Osteosarcoma Xenografts Growth in Mice is Associated with Modulation of Mitochondrial Fission/Fusion Machinery.
Am J Chin Med. 2016; 44(7):1507-1523 [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis  MKI67
MKI67 New Treatment Options for Osteosarcoma - Inactivation of Osteosarcoma Cells by Cold Atmospheric Plasma.
Anticancer Res. 2016; 36(11):5915-5922 [PubMed] Related Publications
MATERIALS AND METHODS: Human osteosarcoma cells (U2-OS and MNNG/HOS) were treated with cold atmospheric plasma and seeded in culture plates. Cell proliferation, p53 and phospho-p53 protein expression and nuclear morphology were assessed.
RESULTS: The treated human osteosarcoma cell lines exhibited attenuated proliferation rates by up to 66%. The cells revealed an induction of p53, as well as phospho-p53 expression, by 2.3-fold and 4.5-fold, respectively, compared to controls. 4',6-diamidino-2-phenylindole staining demonstrated apoptotic nuclear condensation following cold atmospheric plasma treatment.
CONCLUSION: Cold atmospheric plasma treatment significantly attenuated cell proliferation in a preclinical in vitro osteosarcoma model. The resulting increase in p53 expression and phospho-activation in combination with characteristic nuclear changes indicate this was through induction of apoptosis.
Related:
 TP53
TP53 3-Hydroxyflavone inhibits human osteosarcoma U2OS and 143B cells metastasis by affecting EMT and repressing u-PA/MMP-2 via FAK-Src to MEK/ERK and RhoA/MLC2 pathways and reduces 143B tumor growth in vivo.
Food Chem Toxicol. 2016; 97:177-186 [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  MAP2K1
MAP2K1  MMP2
MMP2  Signal Transduction
Signal Transduction  RHOA
RHOA When curing a pediatric tumor is not enough: the case of a psychiatric disorder in a woman surviving osteosarcoma.
Tumori. 2016; 102(Suppl. 2) [PubMed] Related Publications
RESULTS: The high levels of psychological distress and psychopathologic symptoms revealed by this patient prompted us to offer her specific and prolonged support at our institution, since she refused to seek the help of other psychiatric services. The woman revealed a dysfunctional social and family setting and a borderline personality disorder. She was hospitalized after attempting suicide. No psychological distress had previously come to light during her long follow-up for cancer.
CONCLUSIONS: Cancer survivors are at risk of psychological and behavioral problems, so they should be followed up over time. Questionnaires and standard scales are important, but not enough: the physician-patient relationship is crucial to bring out a patient's psychological issues and needs. This means that dedicated resources should be made available, whenever possible.
The clinical significance of changes in ezrin expression in osteosarcoma of children and young adults.
Tumour Biol. 2016; 37(9):12071-12078 [PubMed] Related Publications
Related:
 Bone Cancers
Bone Cancers miR-1 Inhibits Cell Growth, Migration, and Invasion by Targeting VEGFA in Osteosarcoma Cells.
Dis Markers. 2016; 2016:7068986 [PubMed] Free Access to Full Article Related Publications
Related:
 Apoptosis
Apoptosis  Bone Cancers
Bone Cancers  MicroRNAs
MicroRNAs  VEGFA
VEGFA Nanomedicine-based combination of gambogic acid and retinoic acid chlorochalcone for enhanced anticancer efficacy in osteosarcoma.
Biomed Pharmacother. 2016; 83:79-84 [PubMed] Related Publications
Related:
 Apoptosis
Apoptosis miR-10b promotes invasion by targeting KLF4 in osteosarcoma cells.
Biomed Pharmacother. 2016; 84:947-953 [PubMed] Related Publications
METHODS: miR-10b expression in osteosarcoma tissues and osteosarcoma cells were detected using real time PCR. The effects of miR-10b on osteosarcoma cells proliferation, apoptosis, migration and invasion were detected using CCK-8 assay, flow cytometry, wound-healing assay and transwell assay, respectively. The relationship between miR-10b and KLF4 was evaluated using dual-luciferase assay, correlation analysis.
RESULTS: miR-10b was highly expressed in osteosarcoma tissues and osteosarcoma cells. Furthermore, inhibition of miR-10b in osteosarcoma cells depressed the cells proliferation, migration and invasion but promoted cells apoptosis. In addition, KLF4 was down-regulated by miR-10b and miR-10b expression was negatively related to KLF4 expression in osteosarcoma tissue, miR-10b participated in the process of osteosarcoma cells invasion by regulating KLF4 expression.
CONCLUSION: miR-10b is overexpressed in osteosarcoma and KLF4 is the direct target gene of miR-10b. Furthermore, miR-10b promotes osteosarcoma cells progression by downregulating KLF4 expression. These results suggest that miR-10b functions as an oncomiR and play an important role in osteosarcoma cellular processes at least partially through regulating KLF4; miR-10b may be a therapeutic target for osteosarcoma treatment.
Related:
 Apoptosis
Apoptosis  MicroRNAs
MicroRNAs |
Monitor this page |
|
powered by ChangeDetection |
This page last updated: 9th March 2017
Displaying links verified within last 2 weeks at time of update.
